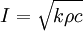To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Volumetric heat capacityVolumetric heat capacity (VHC) describes the ability of a given volume of a substance to store internal energy while undergoing a given temperature change, but without undergoing a phase change. It is different from specific heat capacity in that the VHC depends on the volume of the material, while the specific heat is based on the mass of the material. If given a specific heat value of a substance, one can convert it to the VHC by multiplying the specific heat by the density of the substance. [1] Dulong and Petit predicted in 1818 that ρcp would be constant for all solids (the Dulong-Petit law). In fact, the quantity varies from about 1.2 to 4.5 MJ/m³K. For fluids it is in the range 1.3 to 1.9, and for gases it is a constant 1.0 kJ/m³K. The volumetric heat capacity is defined as having SI units of J/(m³·K). It can also be described in Imperial units of BTU/(ft³·F°). Product highlight
Thermal inertiaThermal inertia is a term commonly used by scientists and engineers modelling heat transfers and is a bulk material property related to thermal conductivity and volumetric heat capacity. For example, this material has a high thermal inertia, or thermal inertia plays an important role in this system, which means that dynamic effects are prevalent in a model, so that a steady-state calculation will yield inaccurate results. The term is a scientific analogy, and is not directly related to the mass-and-velocity term used in mechanics, where inertia is that which limits the acceleration of an object. In a similar way, thermal inertia is a measure of the thermal mass and the velocity of the thermal wave which controls the surface temperature of a material. In heat transfer, a higher value of the volumetric heat capacity means a longer time for the system to reach equilibrium. The thermal inertia of a material is defined as a the square root of the product of the material's bulk thermal conductivity and volumetric heat capacity, where the latter is the product of density and specific heat capacity: SI units of thermal inertia are J m − 2 K − 1 s − 1 / 2 or, equivalently, tiu[2]. For planetary surface materials, thermal inertia is the key property controlling the diurnal and seasonal surface temperature variations and is typically dependent on the physical properties of near-surface geologic materials. In remote sensing applications, thermal inertia represents a complex combination of particle size, rock abundance, bedrock outcropping and the degree of induration. A rough approximation to thermal inertia is sometimes obtained from the amplitude of the diurnal temperature curve (i.e., maximum minus minimum surface temperature). The temperature of a material with low thermal inertia changes significantly during the day, while the temperature of a material with high thermal inertia does not change as drastically. Deriving and understanding the thermal inertia of the surface can help to recognize small-scale features of that surface. In conjunction with other data, thermal inertia can help to characterize surface materials and the geologic processes responsible for forming these materials. Constant volume and constant pressure.For gases it is useful to distinguish between volumetric heat capacity at constant volume and at constant pressure. This distinction has the same meaning as for specific heat capacity. References
See also
Categories: Thermodynamic properties | Thermodynamics | Heat |
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Volumetric_heat_capacity". A list of authors is available in Wikipedia. |