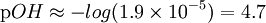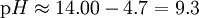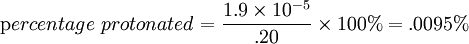To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Weak base
In chemistry, a weak base is a chemical base that does not ionize fully in an aqueous solution. As Bronsted-Lowry bases are proton acceptors, a weak base may also be defined as a chemical base in which protonation is incomplete. This results in a relatively low pH level compared to strong bases. Bases range from a pH of greater than 7 (7 is neutral, like pure water) to 14 (though some bases are greater than 14). The pH level has the formula: Since bases are proton acceptors, the base receives a hydrogen ion from water, H2O, and the remaining H+ concentration in the solution determines the pH level. Weak bases will have a higher H+ concentration because they are less completely protonated than stronger bases and, therefore, more hydrogen ions remain in the solution. If you plug in a higher H+ concentration into the formula, a low pH level results. However, the pH level of bases is usually calculated using the OH- concentration to find the pOH level first. This is done because the H+ concentration is not a part of the reaction, while the OH- concentration is. By multiplying a conjugate acid (such as NH4+) and a conjugate base (such as NH3) the following is given: Since Kw = [H3O + ][OH − ] then, By taking logarithms of both sides of the equation, the following is reached:
Finally, multipying throughout the equation by -1, the equation turns into:
After acquiring pOH from the previous pOH formula, pH can be calculated using the formula pH = pKw - pOH where pKw = 14.00. Weak bases exist in chemical equilibrium much in the same way as weak acids do, with a Base Ionization Constant (Kb) (or the Base Dissociation Constant) indicating the strength of the base. For example, when ammonia is put in water, the following equilibrium is set up: Bases that have a large Kb will ionize more completely and are thus stronger bases. As stated above, the pH of the solution depends on the H+ concentration, which is related to the OH- concentration by the Ionic Constant of water (Kw = 1.0x10-14) (See article Self-ionization of water.) A strong base has a lower H+ concentration because they are fully protonated and less hydrogen ions remain in the solution. A lower H+ concentration also means a higher OH- concentration and therefore, a larger Kb.
Product highlight
Percentage protonatedAs seen above, the strength of a base depends primarily on the pH level. To help describe the strengths of weak bases, it is helpful to know the percentage protonated-the percentage of base molecules that have been protonated. A lower percentage will correspond with a lower pH level because both numbers result from the amount of protonation. A weak base is less protonated, leading to a lower pH and a lower percentage protonated. The typical proton transfer equilibrium appears as such: B represents the base. In this formula, [B]initial is the initial molar concentration of the base, assuming that no protonation has occurred. A typical pH problemCalculate the pH and percentage protonation of a .20 M aqueous solution of pyridine, C5H5N. The Kb for C5H5N is 1.8 x 10-9. First, write the proton transfer equilibrium: The equilibrium table, with all concentrations in moles per liter, is
This means .0095% of the pyridine is in the protonated form of C5H6N+. Examples
Other weak bases are essentially any bases not on the list of strong bases. See also
References
|
|||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Weak_base". A list of authors is available in Wikipedia. |


![\mbox{pH} = -\log_{10} \left[ \mbox{H}^+ \right]](images/math/f/5/2/f527ad48b384c4a6f2328ab8a42a971f.png)
![\mbox{pOH} = -\log_{10} \left[ \mbox{OH}^- \right]](images/math/a/c/7/ac7b52a8f87104b49e536517a397e1df.png)
![K_a \times K_b = {[H_3O^+][NH_3]\over[NH_4^+]} \times {[NH_4^+][OH^-]\over[NH_3]} = [H_3O^+][OH^-]](images/math/1/4/c/14cf0b9c22142396e60185078497a046.png)
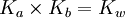
![\mathrm{K_b={[NH_4^+][OH^-]\over[NH_3]}}](images/math/0/e/b/0eb8a1134449c8ec65872d591d4f306a.png)



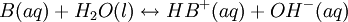
![Percentage\ protonated = {molarity\ of\ HB^+ \over\ initial\ molarity\ of\ B} \times 100\% = {[{HB}^+]\over [B]_{initial}} {\times 100\%}](images/math/4/2/9/429e42c38e12f6306fdfe7023b5bb11c.png)
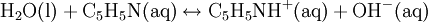
![K_b=\mathrm{[C_5H_5NH^+][OH^-]\over [C_5H_5N]}](images/math/2/e/7/2e78271894ff37cd17623727cf1583f1.png)
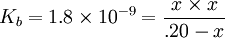
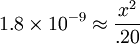
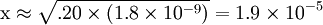
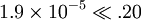 ; so the approximation is valid
; so the approximation is valid