Chemistry for a Raney day
Advertisement
Spanish and Mexican scientists have stumbled across a new way to monoalkylate amines that they claim is more environmentally friendly than alternative routes.
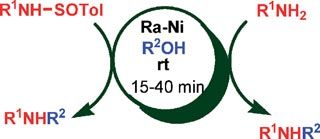
José García Ruano, at the Autonomous University of Madrid, and colleagues were using Raney nickel to cleave nitrogen–sulfur bonds in N-sulfinylamines when they found that changing the solvent produced unexpected products. When they did the reaction in THF, they got primary amines. However, when they changed the solvent to ethanol, they formed N-ethylamines. Although they are not sure of the mechanism, Ruano says he thinks radical intermediates are involved.
Ruano showed that a variety of primary and secondary alcohols can be used to alkylate the amines. Also, he found that dialkylation, a common problem in amine alkylation reactions, does not occur. Ruano recovered the Raney nickel by filtration after the reactions and demonstrated that it could be reused.
‘Many of the methods [for alkylating amines] reported so far are undesirable from an environmental point of view, due to the generation of wasteful byproducts,’ says Ruano. ‘The broad scope of this method, which does not generate byproducts, along with the heterogeneous character of the catalyst suggests it is one of the most convenient methods reported so far.’
Ruano says that the method could be used on an industrial scale because the catalyst is inexpensive and easily recovered. He is now working on monoalkylating amino acids and peptides and also tandem processes where he reduces imines then alkylates them.
Original publication: José Luis García Ruano et al., Chem. Commun., 2009.