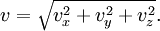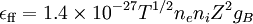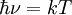To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
BremsstrahlungBremsstrahlung (pronounced [ˈbʁɛmsˌʃtʁaːlʊŋ] , from German bremsen "to brake" and Strahlung "radiation", i.e. "braking radiation" or "deceleration radiation"), is electromagnetic radiation produced by the deceleration of a charged particle, such as an electron, when deflected by another charged particle, such as an atomic nucleus. The term is also used to refer to the process of producing the radiation. Bremsstrahlung has a continuous spectrum. The phenomenon was discovered by Nikola Tesla during high frequency research he conducted between 1888 and 1897. Bremsstrahlung may also be referred to as free-free radiation. This refers to the radiation that arises as a result of a charged particle that is free both before and after the deflection (acceleration) that causes the emission. Strictly speaking, bremsstrahlung refers to any radiation due to the acceleration of a charged particle, which includes synchrotron radiation; however, it is frequently used (even when not speaking German) in the more narrow sense of radiation from electrons stopping in matter. The word Bremsstrahlung is retained from the original German to describe the radiation which is emitted when electrons are decelerated or "braked" when they are fired at a metal target. Accelerated charges give off electromagnetic radiation, and when the energy of the bombarding electrons is high enough, that radiation is in the x-ray region of the electromagnetic spectrum. It is characterized by a continuous distribution of radiation which becomes more intense and shifts toward higher frequencies when the energy of the bombarding electrons is increased.
Product highlight
Outer"Outer bremsstrahlung" is the term applied in cases where the energy loss by radiation greatly exceeds that by ionization as a stopping mechanism in matter. This is seen clearly for electrons with energies above 50 keV. Inner"Inner bremsstrahlung" is the term applied to the less frequent case of radiation emission during beta decay, resulting in the emission of a photon of energy less than or equal to the maximum energy available in the nuclear transition. Inner bremsstrahlung is caused by the abrupt change in the electric field in the region of the nucleus of the atom undergoing decay, in a manner similar to that which causes outer bremsstrahlung. In electron and positron emission the photon's energy comes from the electron/neutron pair, with the spectrum of the bremsstrahlung decreasing continuously with increasing energy of the beta particle. In electron capture the energy comes at the expense of the neutrino, and the spectrum is greatest at about one third of the normal neutrino energy, reaching zero at zero energy and at normal neutrino energy. Beta particle emitting substances sometimes exhibit a weak radiation with continuous spectrum that is due to both outer and inner bremsstrahlung, or to one of them alone. Secondary radiationBremsstrahlung is a type of "secondary radiation", in that it is produced as a result of stopping (or slowing) the primary radiation (beta particles). In some cases, e.g. 32P, the Bremsstrahlung produced by shielding this radiation with the normally used dense materials (e.g. lead) is itself dangerous; in such cases, shielding must be accomplished with low density materials, e.g. Plexiglas, Lucite, plastic, wood, or water [1]; because the rate of deceleration of the electron is slower, the radiation given off has a longer wavelength and is therefore less penetrating. The case where acceleration is parallel to velocityIf a particle of charge q experiences an acceleration
Integration by parts then gives the total power emitted as
Note that, since From a plasma
In a plasma the free electrons are constantly producing Bremsstrahlung in collisions with the ions. The power spectral density (power per angular frequency interval per volume, integrated over all solid angle) of the Bremsstrahlung radiated, is calculated to be [2]
ns is the number density of species s (s = e,Z for electrons, ion species Z), Te is the electron temperature in energy units, and symbols not defined here are physical constants. Note that the third bracketed factor on the right-hand side determines its units. The "effective" ion charge state Zeff is given by an average over the charge states of the ions:
The special function E1 is defined in the exponential integral article, and
(km is a maximum or cutoff wavenumber). km = K / λB when Te > Z2 27.2 eV (for a single ion species; 27.2 eV is twice the ionization energy of hydrogen) where K is a pure number and For the case km = K / λB, we find
dPBr / dω is infinite at ω = 0, and decreases rapidly with ω. The resulting power density, integrated over all frequencies, is finite and equals
Note the appearance of the fine structure constant α due to the quantum nature of λB. In practical units, a commonly used version of this formula is [3]
This formula agrees with the theoretical estimate if we set K=3.17; the value K=3 is suggested by Ichimaru. For very high temperatures there are relativistic corrections to this formula, that is, additional terms of order Te/mec2.[2] In astrophysicsThe dominant luminous component in a cluster of galaxies is the 107 to 108 Kelvin intracluster medium. The emission from the intracluster medium is characterized by thermal Bremsstrahlung. Thermal Bremsstrahlung radiation is when the particles populating the emitting plasma are at a uniform temperature and are distributed according to the Maxwell–Boltzmann distribution where speed, v, is defined as The bulk emission from this gas is thermal Bremsstrahlung. The power emitted per cubic centimeter per second can be written in the compact form with cgs units [erg cm-3 s-1] and where 'ff' stands for free-free, 1.4x10-27 is the condensed form of the physical constants and geometrical constants associated with integrating over the power per unit area per unit frequency, ne and ni are the electron and ion densities, respectively, Z is the number of protons of the bending charge, gB is the frequency averaged Gaunt factor and is of order unity, and T is the global x-ray temperature determined from the spectral cut-off frequency below which no photons are created because the energy supplied by acceleration of the electron by the positively charged nucleus is less than the minimum energy required for creation of a photon. This process is also known as Bremsstrahlung cooling since the plasma is optically thin to photons at these energies and the energy radiated is emitted freely into the universe. This radiation is in the energy range of X-rays and can be easily observed with space-based telescopes such as Chandra X-ray Observatory, XMM-Newton, ROSAT, ASCA, EXOSAT, Astro-E2, and future missions like Con-X and NeXT[3].
References
See also
Categories: Atomic physics | Plasma physics | Scattering |
|||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Bremsstrahlung". A list of authors is available in Wikipedia. |





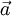 which is collinear with its velocity
which is collinear with its velocity 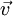 , the angular distribution of the bremsstrahlung is
, the angular distribution of the bremsstrahlung is
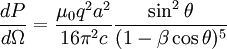 ,
,
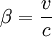 and
and 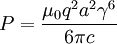 ,
,
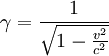 is the Lorentz factor.
is the Lorentz factor.
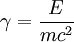 , for a given energy E,
, for a given energy E, 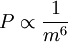 . So, if an
. So, if an ![{dP_\mathrm{Br} \over d\omega} = {4\sqrt 2 \over 3\sqrt\pi} \left[ n_er_e^3 \right]^2 \left[ { m_ec^2 \over T_e } \right]^{1/2} \left[ {m_ec^2 \over r_e^3} \right] Z_\mathrm{eff} E_1(w_m)](images/math/8/d/d/8ddcb58430ac97696e89f41d3a2dac6f.png)
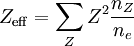
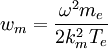
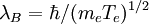 is a thermal electron
is a thermal electron 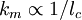 where
where ![w_m = {1 \over 2K^2} \left[{\hbar\omega \over T_e}\right]^2](images/math/9/a/c/9ac0de53375bb309eddb225afafcc058.png)
![P_\mathrm{Br} = {8 \over 3} \left[ n_er_e^3 \right]^2 \left[ { T_e \over m_ec^2} \right]^{1/2} \left[ {m_ec^3 \over r_e^4} \right] Z_\mathrm{eff} \alpha K](images/math/0/e/6/0e61310fc91aa809344f7f973cecab18.png)
![P_\mathrm{Br} [\textrm{Watt/m}^3] = \left[{n_e \over 7.69 \times 10^{18} \textrm{m}^{-3} }\right]^2 T_e[\textrm{eV}]^{1/2} Z_\mathrm{eff}](images/math/2/7/e/27ecc0442fd5068df14ee2fd423768ae.png)
![f (v) = 4 \pi \left(\frac{m}{2 \pi kT}\right)^{3/2} v^2 \exp \left[ \frac{-mv^2}{2kT} \right]](images/math/6/b/7/6b75e3db8fd0a77fb94ce48cb22bb619.png)