To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Electronic correlationElectronic correlation refers to the interaction between electrons in a quantum system whose electronic structure is being considered. The term correlation stems from mathematical statistics and means that two distribution functions, f and g, are not independent of each other. Product highlight
Atomic and Molecular Systems
Within the Hartree-Fock method of quantum chemistry, the antisymmetric wave function is approximated by a single Slater determinant. Exact wave functions, however, cannot generally be expressed as single determinants. The single-determinant approximation does not take into account Coulomb correlation, leading to a total electronic energy different from the exact solution of the non-relativistic Schrödinger equation within the Born-Oppenheimer approximation. Therefore the Hartree-Fock limit is always above this exact energy. The difference is called the correlation energy, a term coined by Löwdin. A certain amount of electron correlation is already considered within the HF approximation, found in the electron exchange term describing the correlation between electrons with parallel spin. This basic correlation prevents two parallel-spin electrons from being found at the same point in space and is often called Fermi correlation. Coulomb correlation, on the other hand, describes the correlation between the spatial position of electrons with opposite spin due to their Coulomb repulsion. There is also a correlation related to the overall symmetry or total spin of the considered system. In simple terms the molecular orbitals of the Hartree-Fock method are optimised by evaluating the energy of an electron in each molecular orbital moving in the mean field of all other electrons, rather than including the instantaneous repulsion between electrons. Crystalline SystemsIn condensed matter physics, electrons are typically described with reference to a periodic lattice of atomic nuclei. Non-interacting electrons are therefore typically described by Bloch waves, which correspond to the delocalized, symmetry adapted molecular orbitals used in molecules (while Wannier functions correspond to localized MOs). A number of important theoretical approximations have been proposed to explain electron correlations in these crystalline systems. The Fermi liquid model of correlated electrons in metals is able to explain the temperature dependence of resistivity by electron-electron interactions. Superconductivity is the result of electron correlations. The Hubbard model model is based on the tight-binding approximation, and can explain conductor-insulator transitions in Mott insulators such as transition metal oxides by the presence of repulsive Coulombic interactions between electrons. The RKKY Interaction can explain electron spin correlations between unpaired inner shell electrons in different atoms in a conducting crystal by a second-order interaction that is mediated by conduction electrons. The Tomonaga Luttinger Liquid model approximates second order electron-electron interactions as bosonic interactions. Mathematical viewpointFor two independent electrons a and b,
where ρ(ra,rb) represents the joint electronic density, or the probability density of finding electron a at ra and electron b at rb. Within this notation, ρ(ra,rb)dradrb represents the probability of finding the two electrons in their respective volume elements dra and drb. If these two electrons are correlated, then the probability of finding electron a at a certain position in space depends on the position of electron b, and vice versa. In other words, the product of their independent density functions does not adequately describe the real situation. At small distances, the uncorrelated pair density is too large; at large distances, the uncorrelated pair density is too small (i.e. the electrons tend to "avoid each other"). See also
Categories: Atomic physics | Quantum chemistry | Electron |
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Electronic_correlation". A list of authors is available in Wikipedia. |





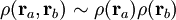 ,
,


