Breakthrough: Novel technique seamlessly converts ammonia to green hydrogen
Findings have attracted significant attention from academic research communities
Advertisement
A research team, led by Professor Guntae Kim in the School of Energy and Chemical Engineering at UNIST has announced a breakthrough in technology that efficiently converts liquid ammonia into hydrogen. Their findings have also attracted significant attention from academic research communities owing to its new analysis protocol, capable of finding optimal process environments.
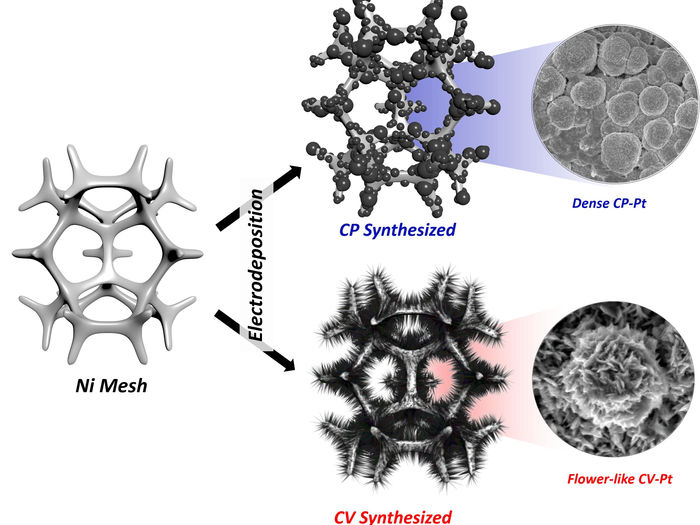
Schematic illustration of the CP-Pt and CV-Pt electrodes.
UNIST
In this study, the research team succeeded in producing green hydrogen (H2) in large quantities with a purity of nearly 100 percent by decomposing liquid ammonia (NH3) into electricity. Besides, according to the research team, such method consumed three times less power than hydrogen made using electrolysis of water.
Ammonia has emerged as an attractive potential hydrogen carrier due to its extremely high energy density, and ease of storage and handling. Moreover, the electrolysis of ammonia to produce nitrogen and hydrogen only requires an external voltage of 0.06 V theoretically, which is much lower than the energy needed for water electrolysis (1.23 V), noted the research team.
In this study, the research team propose a well-established procedure using in operando gas chromatography that enables us to reliably compare and evaluate the new catalyst for ammonia oxidation. According to the research team, with the protocol, they could distinguish in detail the competitive oxidation reaction between the ammonia oxidation and oxygen evolutionreactions with real-time monitoring.
With the use of flower-like electrodeposited Pt catalyst, researchers have efficiently produced hydrogen with less power consumption of 734 LH2 kW h−1, which is significantly lower than that of the water-splitting process (242 LH2 kW h−1). “The use of this rigorous protocol should help to evaluate the practical performances for ammonia oxidation, thus enabling the field to focus on viable pathways towards the practical electrochemical oxidation of ammonia to hydrogen,” noted the research team.
Most read news
Other news from the department science

Get the chemical industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.