Improve industrial chemical reactions with better products and less byproducts
Heterogeneous photocatalysis can help scientists sustainably control products and byproducts of chemical reactions, including reducing CO₂
Advertisement
Controlling the products and byproducts of a chemical reaction is called selectivity. Scientists are trying to find better ways to control selectivity in industrial chemical reactions to improve the quality of the chemical products, reduce wasteful byproducts, and reduce harmful byproducts, like the emission of useless CO2. There are various catalytic modes that scientists can use in chemical reactions. Catalytic modes include heterogeneous, homogenous, and biological catalysis, but heterogeneous catalysis has many advantages over the others, because it can be operated continuously, and it is more easily scalable. Researchers set out to find if it also can help improve selectivity in industrial chemical reactions.
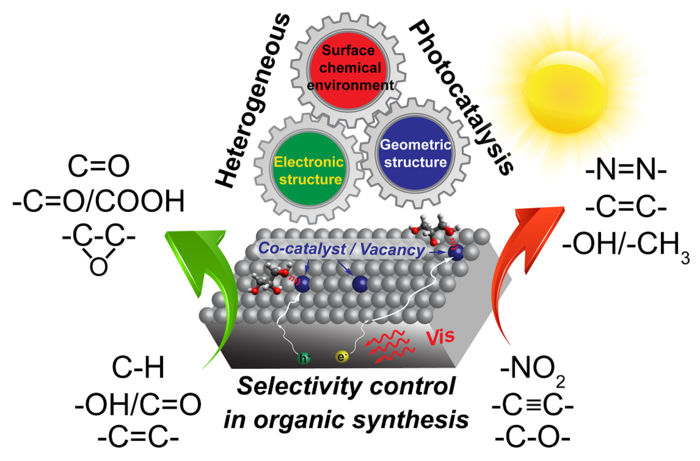
Researchers demonstrated how products and byproducts of chemical reactions could be sustainably and easily controlled by applying heterogeneous photocatalysis.
Nano Research Energy
By applying heterogeneous photocatalysis, researchers demonstrated how products and byproducts of chemical reactions could be sustainably and easily controlled. Heterogeneous photocatalysis is the use of light in chemical reactions, where the catalyst and the reactor are in different phases. This technique is especially interesting to researchers because the conditions required are mild.
The technique and its implications for selectivity in organic synthesis were described in a paper published on May 12 in Nano Research Energy.
“No matter what kinds of catalytic systems are employed, one key evaluation indicator always dominates, especially in the field of organic synthesis: the selectivity of desired products. High selectivity not only provides the high purity of final products, but it also reduces the cost of removing byproducts,” said paper author Yitao Dai, a professor at the Suzhou Institute for Advanced Research at the University of Science and Technology of China in Suzhou, China. “In contrast, undesired overoxidation or overreduction leads to low-value products, coke deposition, or even mineralization with the emission of useless CO2. Therefore, it is one main and important scientific subject in organic chemistry how to precisely control selectivity.”
Researchers demonstrated how heterogeneous photocatalysis can improve various organic transformations across industries. For example, the pulp and paper industry is a well-known polluter and one cause of the pollution is a byproduct called lignin. Around 50 million tons of lignin-containing byproducts are produced by the pulp and paper industry per year and only 2% of that can be recovered and used again. Researchers demonstrated how heterogeneous photocatalysis can break down lignin in mild conditions.
Reducing the environmental cost of industrial chemical reactions is an important motivator for this kind of research. “Given the current global theme of development in CO2 reduction and sustainable energy utilization, heterogeneous photocatalysis can act as one promising protocol to control selectivity in organic transformations with reduced energy consumption,” said Dai. “It enables sustainable solar-to-chemical energy conversion under mild conditions and offers unique reaction pathways for improved selectivity control.” One benefit of using heterogeneous photocatalysis is the mild conditions that can be used to complete the chemical reactions. These include using air as the oxidant, mild room temperature, and utilizing ambient pressure. This makes it a simpler technique to use when compared to others.
Looking ahead, researchers are thinking about the implications of applying this technique broadly. The researchers’ ultimate goal is to create an artificial photosynthetic plant. This would allow the sustainable, continuous production of organic compounds using heterogeneous photocatalytic systems. This method would be cheap, effective, and utilize sunlight. In the meantime, though, more research needs to be done. “For the field of organic synthesis via heterogeneous photocatalysis, more and more research focus should be on two aspects. First, existing challenging reactions. Second, new organic reactions,” Dai said. “We expect that the design of novel reaction routes based on the catalytic properties of photocatalysts may provide a solution for some quite challenging organic syntheses, generating products with high regioselectivity or asymmetry.”

































































