Nanoparticles drill holes at will in silicon
Industrially usable technique for producing porous silicon and glass opens up fresh perspectives for numerous applications
Advertisement
A Hamburg-based research team has developed a new method for producing a network of large to very fine pores in silicon and glass. At DESY’s X-ray source PETRA III, scientists led by DESY researchers Stella Gries and Patrick Huber were able to analyse the porosity using x-ray tomography. The method for producing micro- and nanochannels, which the team is presenting in the journal Small, is easy to control, extremely variable and even works for large volumes of materials. This makes it ideal for use in industrial applications.
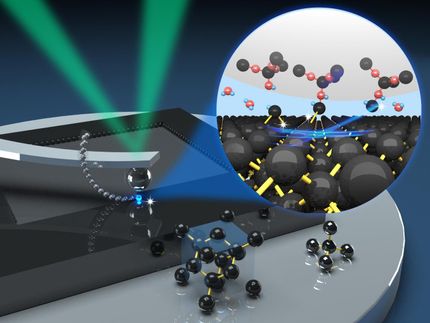
Silver nanoparticles are able to drill nanochannels in macroporous silicon and thus to hierarchically porosify silicon on the wafer scale. The graphics presents the real porous material combined with an illustration of the porosification process, i.e. the chemical etching and thus pore formation along the trajectories of self-propelled, catalytically acting silver particles under the development of hydrogen bubbles in aqueous solution.
DESY/TUHH, AG Huber
In road networks, the method is common practice: while large motorways provide fast connections for long-distance traffic, small roads and lanes can be used to reach even the remotest corners of the country – albeit much more slowly. Nature often does the same thing, for example in the lungs or in plants: bronchial tubes or networks of large capillaries in leaves allow air or water to be transported rapidly across tissues, and then the channels branch out and become narrower until they reach the tiny alveoli or pores, where important local functions take place, such as supplying oxygen to the blood or carrying out photosynthesis. These highly efficient structures are known as hierarchical porous systems, and – being divided into large and small units – they can take care of extensive distribution or local functions.
The research team led by DESY PhD student Gries is now able to produce silicon crystals with precisely such hierarchical porous systems. To achieve this, the scientists coated silicon wafers, which already had straight channels one micrometre in diameter passing through them, with silver nanoparticles. The 20 to 60 nanometre particles are deposited on the surface of the wafer. The scientists then exposed the wafers to a corrosive solution of hydrofluoric acid and hydrogen peroxide, triggering a fascinating and, at first glance, unbelievable process: the nanoparticles bore into the silicon, dissolving the silicon crystal at the surfaces where the silver particle was in contact with the silicon. Just as in the once popular computer game Pac-Man, the silver particles burrow further and further into the solid, leaving behind a fine network of tunnels along the way. The kinetic energy for the directed propulsion of the particles comes from a chemical decomposition reaction, i.e. the conversion of the hydrogen peroxide into water and hydrogen and the “eating up” of the silicon. The silver particles behave like small, autonomous Pac-Man agents, catalysing the reaction and thus enabling their own propulsion through the silicon crystal. The resulting system of tunnels self-organises to create the desired three-dimensional hierarchical porous structure.
The research team used a number of different methods to investigate the porosity of the silicon. With the help of X-ray tomographic images taken at the PETRA III beamline P05, they were able to resolve the inner structure on a nanometre scale. “We see that the etching process systematically perforates the entire crystal, creating nanopores that are smaller than 100 nanometres,” says principal author Gries, who developed this method in Patrick Huber’s group in the course of her master’s thesis and is now conducting further research as part of her doctorate. “By adjusting the size of the nanoparticles and the duration of the procedure, we can precisely control how deep the hierarchical porous system extends,” adds Manuel Brinker, a member of the group that helped supervise Stella Gries’s research. Longer exposures produce pores that penetrate to the parallel main channels in the silicon wafer, connecting them with each other. For the time being, the exact mechanisms that lead to the movement of the particles, and thus to the formation of the network of channels, are only partially understood. Sometimes, for example, the particles travel along spiral paths, leading to spiral-shaped nanochannels in the silicon, and sometimes they change direction abruptly, suggesting that the particles are rotating. The network of pores produced has a sponge-like structure and thus no preferred orientation, unlike the large main channels. “We suspect that the geometric shape of the silver nanoparticles has a strong influence on the way the particles eat into the silicon,” says Gries.
In a further step, the scientists heated the perforated silicon to over 800 degrees Celsius in an atmosphere containing oxygen. The walls between the tunnels are so thin that the silicon was completely oxidised to form silicon dioxide, colloquially known as glass. To the scientists’ great surprise, the structure of channels was not destroyed despite the considerable rearrangement of the atoms and the expansion of the walls when the oxygen was incorporated. This means that the open-pored structure allows the wafers to be transformed into hierarchical porous glass. The team was able to make this material, which has a milky appearance due to the way its pores reflect white light, transparent by infiltrating it with water. This means the amount of light absorbed by the glass can be very easily controlled by moistening and drying the material, an effect that could be used, for example, for simple functions in windows, which could be switched by the humidity in the air. Such smart glasses could be switched comparatively quickly because wetting and drying can take place quickly throughout the entire volume thanks to the multiscale transportation routes.
Overall, the scientists anticipate a wide range of potential applications, including in energy technology. “Silicon still has the highest potential to serve as an electrode material for lithium-ion batteries,” says Patrick Huber (DESY and TU Hamburg). “Our new etching technique may form the basis for a new generation of battery cells with a high charge density and a large number of charging cycles, if it turns out that not only vitrification, i.e. the incorporation of oxygen, but also the incorporation of lithium preserves the internal structure thanks to the hierarchical porosity of the silicon crystals. In non-porous silicon, this lithiation usually destroys the material.”
The next step will be to gain an even better understanding of how the manufacturing parameters affect porosity and what exactly drives forward the silver particles as they are perforating the material. Stella Gries intends to look into this in her doctorate. In a next step, we will also be investigating the lithiation of the hierarchical porous silicon in cooperation with other partners.
The procedure, for which the researchers have applied for a patent, was developed within the framework of the Centre for Integrated Multiscale Materials Systems, CIMMS, and the Collaborative Research Centre “SFB 986: Tailor-Made Multiscale Materials Systems”, which is based at the Hamburg University of Technology.