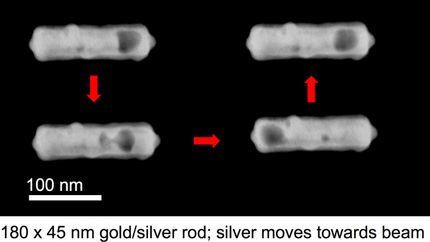
Surface modification of gold nanorods inside polymer beads
'Gold nanorods can be made stable and soluble in both polar and non-polar solvents', say US scientists
Advertisement
A team led by Qun Huo at the University of Central Florida, Orlando, have developed a method to replace the ionic surfactant layer on a gold nanorod surface with a more stable thiol ligand. This is carried out by a place exchange reaction conducted inside an ionic exchange polymer bead.
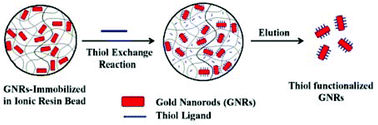
The gold nanorods were first loaded into porous ionic exchange polymer beads. The trapped gold nanorods were then exchanged for a bifunctional thiol ligand inside the polymer beads leading to successful surface modification of the gold nanorods with a thiol ligand protecting layer. This process is called a place exchange reaction.
Huo explains that place exchange reactions in solution are often problematic when applied to gold nanorods. This is because such direct modification in solution usually leads to an irreversible agglomeration of the gold nanorods. Huo's method prevents this by controlling the thiol ligand place exchange reaction inside the pores of a polymer bead. 'The trapping of gold nanorods inside polymer beads temporarily allows sufficient time for a stable thiol monolayer to form on the gold nanorod surface,' adds Huo.
The resulting gold nanorods show good solubility and stability against aggregation in both polar and non-polar solvents. These types of gold nanorods are known to possess many interesting optical properties and have potential application in biological systems.
Original publication: Qiu Dai, Janelle Coutts, Jianhua Zou and Qun Huo; Chem. Commun., 2008.