AAC Infotray
Laboratory information management systems:
Limsophy
Moving Data: Across Systems, Technologies and Time

The laboratory of today is overflowing with data. Powerful laboratory information management systems (LIMS) are needed to keep operations efficient. A LIMS needs to be able to move data across systems, technologies and time for the value of the investment to last.
Systems:
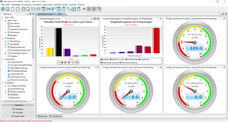
A high level of administrative effort in the laboratory indicates a need for better data management. Moving data across different systems is inefficient. This should only have to happen when migrating data while first configuring an integrated system. Limsophy LIMS supports this central processing with the highest level of user convenience and greatly reduces administration costs. Peripheral processes are optimally integrated through interfaces, which the customers can adjust by themselves.
Technologies:
Technologies are subject to change over time, which means that data must be moved. We understand technologies to be the underlying principles on which software products or processes are based. Limsophy LIMS lets you easily transfer data from different database versions and technologies into new database versions and technologies.
Time:
Work processes change over time. Limsophy offers what you need to integrate modifications to work processes into the software. Modified work processes, new systems or technologies may require data to be moved over time. Two strategies have been established:
1) Outdated data formats are converted into new data formats. Limsophy LIMS helps to automate this process.
2) The data accessibility is maintained by the data being stored in "timeless" formats (e. g. tiff, txt, ASCII or xml) not based on a particular software version. The downside is that you have to compromise on convenience in the data collection.
Intelligently Cover Laboratory Requirements
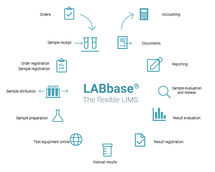
Limsophy products cover the essential requirements of all types of laboratories: Limsophy LIMS for analytical and measuring laboratories, Limsophy RIMS for development labs and Limsophy RALIMS for analytical and R&D laboratories.
The complementary products Limsophy Inspection for planning and carrying out inspections and audits, Limsophy Creation for formulation development and Limsophy BPM for the maintenance of quality management systems complete the offer.
Limsophy Creation
- Targeted development of formulations
- The unique blend of formulation and analysis in software generates additional knowledge
- The combined search for formulation properties and analytical results opens up new possibilities
- Convenient management of raw materials, intermediate and end products
- Permanent knowledge preservation through database support
Limsophy Inspection
- Mobile inspection reviews
- Planning and automatic determination of (post)-inspection dates through risk-based review of operations
- Creating checklists and reports
- Easy management of licenses, orders and complaints
Limsophy RALIMS
- Combines the world of ELN and LIMS
- Ideal for research and complex project structures
- Centralized management of laboratory data and results
- Detailed, flexible description of tests
- Trial information and research results still fully available even after years
- Efficient, regular production of reports using templates
Limsophy BPM
- Simple, sleek quality management, immediately comprehensible for everyone
- Automatic generation of flowcharts, including responsibilities and applicable documents
- Simple relationships between processes, functions and people
- Automated document control
- Personnel management with function matrix, representative regulation and training management
- Integrated audit, objectives and risk management, including a continuous improvement process
Product classification Limsophy
Product categories
Applications
Manufacturers of similar products
Find more laboratory information management systems and related products
Find Limsophy and related products in the theme worlds
Topic world Digitalization in the laboratory
The topic world Digitalization in the lab presents innovations and trends from digital data systems (ELN, LIMS) to laboratory robots and networked devices (IoT) to AI and machine learning.

Topic world Digitalization in the laboratory
The topic world Digitalization in the lab presents innovations and trends from digital data systems (ELN, LIMS) to laboratory robots and networked devices (IoT) to AI and machine learning.