GUS LAB
Laboratory information management systems:
LABS/Q LIMS
LABS/Q LIMS - The professional standard solution for all laboratories
GUS LAB GmbH
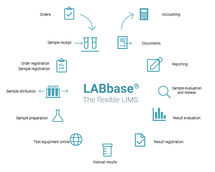
LABS/Q is a state-of-the-art laboratory information and management system (LIMS) that has been used by over 400 customers worldwide for more than 30 years. The extensive functionality of LABS/Q allows implementations to remain very close to the standard and reduces customization to a minimum. Customers from industries such as pharmaceuticals, chemicals, contract laboratories, food, and many more appreciate the robustness, performance, and flexibility of LABS/Q.
With LABS/Q, you can manage all your laboratory data in one application and only need to enter the required master data. If you already have data in another system, we will migrate this data using an automated process. When implementing LABS/Q, you benefit from lower costs because the introduction phase is designed to be efficient. In addition, project times are shortened, which means you can reap the benefits faster. Easy and risk-free upgradeability.
Product classification LABS/Q LIMS
Product categories
Applications
Manufacturers of similar products
Find more laboratory information management systems and related products
Find LABS/Q LIMS and related products in the theme worlds
Topic world Digitalization in the laboratory
The topic world Digitalization in the lab presents innovations and trends from digital data systems (ELN, LIMS) to laboratory robots and networked devices (IoT) to AI and machine learning.

Topic world Digitalization in the laboratory
The topic world Digitalization in the lab presents innovations and trends from digital data systems (ELN, LIMS) to laboratory robots and networked devices (IoT) to AI and machine learning.