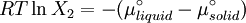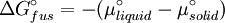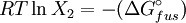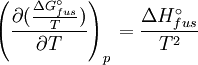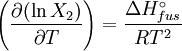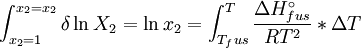To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Enthalpy of fusionThe standard enthalpy of fusion (symbol: ΔHfus), also known as the heat of fusion or specific melting heat, is the amount of thermal energy which must be absorbed or evolved for 1 mole of a substance to change states from a solid to a liquid or vice versa. It is also called the latent heat of fusion or the enthalpy change of fusion, and the temperature at which it occurs is called the melting point. When you withdraw thermal energy from a liquid or solid, the temperature falls. When you add heat energy the temperature rises. However, at the transition point between solid and liquid (the melting point), extra energy is required (the heat of fusion). To go from liquid to solid, the molecules of a substance must become more ordered. For them to maintain the order of a solid, extra heat must be withdrawn. In the other direction, to create the disorder from the solid crystal to liquid, extra heat must be added. The heat of fusion can be observed if you measure the temperature of water as it freezes. If you plunge a closed container of room temperature water into a very cold environment (say −20 °C), you will see the temperature fall steadily until it drops just below the freezing point (0 °C). The temperature then rebounds and holds steady while the water crystallizes. Once completely frozen, the temperature will fall steadily again. The temperature stops falling at (or just below) the freezing point due to the heat of fusion. The energy of the heat of fusion must be withdrawn (the liquid must turn to solid) before the temperature can continue to fall. The units of heat of fusion are usually expressed as:
Product highlight
Reference values of common substances
These values are from the CRC Handbook of Chemistry and Physics, 62nd edition. The conversion between cal/g and kJ/kg in the above table uses the thermochemical calorie (calth) = 4.184 joules rather than the International Steam Table calorie (calINT) = 4.1868 joules. ApplicationsTo heat one kilogram (about 1 litre) of water from 10 °C to 30 °C requires 20 kcal.
Solubility predictionThe heat of fusion can also be used to predict solubility for solids in liquids. Provided an ideal solution is obtained the mole fraction of solute at saturation is a function of the heat of fusion, the melting point of the solid and the temperature of the solution: For example the solubility of paracetamol in water at 298 K is predicted to be: This equals to a solubility in grams per liter of:
which is a deviation from the real solubility (240 g/L) of 11%. This error can be reduced when an additional heat capacity parameter is taken into account [1] ProofAt equilibrium the chemical potentials for the pure solvent and pure solid are identical: or
Rearranging gives: and since the heat of fusion being the difference in chemical potential between the pure liquid and the pure solid, it follows that Application of the Gibbs-Helmholtz equation: ultimately gives: or: and with integration:
the end result is obtained: See also
References
|
||||||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Enthalpy_of_fusion". A list of authors is available in Wikipedia. |





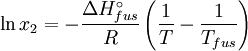
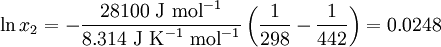
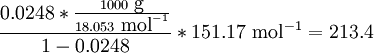
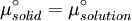
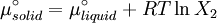
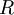 the
the 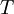 the
the