To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Dalton's law
Product highlightMathematically, the pressure of a mixture of gases can be defined as the summation
It is assumed that the gases do not react with each other. where The relationship below provides a way to determine the volume based concentration of any individual gaseous component. where: Dalton's law is not exactly followed by real gases. Those deviations are considerably large at high pressures. In such conditions, the volume occupied by the molecules can become significant compared to the free space between them. Moreover, the short average distances between molecules raises the intensity of intermolecular forces between gas molecules enough to substantially change the pressure exerted by them. Neither of those effects are considered by the ideal gas model. See also
Categories: Distillation | Gas laws | Physical chemistry | Chemical engineering |
||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Dalton's_law". A list of authors is available in Wikipedia. | ||||||||||





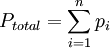 or
or 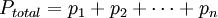
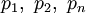 represent the partial pressure of each component.
represent the partial pressure of each component.
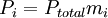
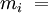 the
the 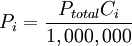
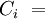 is the concentration of the ith component expressed in
is the concentration of the ith component expressed in 

