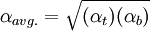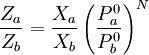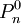To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Fenske equationThe Fenske equation in continuous fractional distillation is an equation used for calculating the minimum number of theoretical plates required for the separation of a binary feed stream by a fractionation column that is being operated at total reflux (i.e., which means that no overhead product distillate is being withdrawn from the column). Product highlightThe equation was derived by Merrell Fenske in 1932 [1], a professor who served as the head of the chemical engineering department at the Pennsylvania State University from 1959 to 1969. This is one of the many different but equivalent versions of the Fenske equation:[2][3][4][5]
For ease of expression, the more volatile and the less volatile components are commonly referred to as the light key (LK) and the heavy key (HK), respectively. If the relative volatility of the light key to the heavy key is constant from the column top to the column bottom, then αavg. is simply α. If the relative volatility is not constant from top to bottom of the column, then the following approximation may be used:[2]
The above Fenske equation can be modified for use in the total reflux distillation of multi-component feeds.[3] Another form of the Fenske equationA derivation of another form of the Fenske equation for use in gas chromatography is available on the U.S. Naval Academy's web site.[6] Using Raoult's law and Dalton's Law for a series of condensation and evaporation cycles (i.e., equilibrium stages), the following form of the Fenske equation is obtained:
See also
References
Categories: Distillation | Chemical engineering | Separation processes |
||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Fenske_equation". A list of authors is available in Wikipedia. | ||||||||||||||||||||||||||||||||||||





![\ N = \frac{\log \, \bigg[ \Big(\frac{X_d}{1-X_d}\Big)\Big(\frac{1-X_b}{X_b} \Big) \bigg]}{\log \, \alpha_{avg}}](images/math/8/4/2/842a9616e370ef4db3164f048f797917.png)