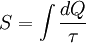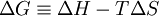To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Entropy and lifeMuch writing and research has been devoted to the relationship between the thermodynamic quantity entropy and the evolution of life. In 1910, American historian Henry Adams printed and distributed to university libraries and history professors the small volume A Letter to American Teachers of History proposing a theory of history based on the second law of thermodynamics and the principle of entropy.[1][2] The 1944 book What is Life? by Nobel-laureate physicist Erwin Schrödinger served largely to stimulate this research. In this book, Schrödinger states that life feeds on negative entropy, or negentropy as it is sometimes called. Recent writings have utilized the concept of Gibbs free energy to elaborate on this issue. Product highlight
Origin
In 1863, Rudolf Clausius published his noted memoir "On the Concentration of Rays of Heat and Light, and on the Limits of its Action" wherein he outlined a preliminary relationship, as based on his own work and that of William Thomson, between his newly developed concept of entropy and life. Building on this, one of the first to speculate on a possible thermodynamic perspective of evolution was the Austrian physicist Ludwig Boltzmann. In 1875, building on the works of Clausius and Kelvin, Boltzmann reasoned[3]:
Early viewsIn 1876, American civil engineer Richard Sears McCulloch, in his Treatise on the Mechanical Theory of Heat and its Application to the Steam-Engine, which was an early thermodynamics textbook, states, after speaking about the laws of the physical world, that "there are none that are established on a firmer basis than the two general propositions of Joule and Carnot; which constitute the fundamental laws of our subject." McCulloch then goes on to show that these two laws may be combined in a single expression as follows: where
McCullen then declares that the applications of these two laws, i.e. what are presently known as the first law of thermodynamics and the second law of thermodynamics, are innumerable. He then states:
McCulloch then gives a few examples of what he calls the “more interesting examples” of the application of these laws in extent and utility. The first example he gives, is physiology wherein he states that “the body of an animal, not less than a steamer, or a locomotive, is truly a heat engine, and the consumption of food in the one is precisely analogous to the burning of fuel in the other; in both, the chemical process is the same: that called combustion.” He then incorporates a discussion of Lavoisier’s theory of respiration with cycles of digestion and excretion, perspiration, but then contradicts Lavoisier with recent findings, such as internal heat generated by friction, according to the new theory of heat, which, according to McCullen, states that the “heat of the body generally and uniformly is diffused instead of being concentrated in the chest”. McCullen then gives an example of the second law, where he states that friction, especially in the smaller blooded-vessels, must develop heat. Without doubt, animal heat is thus in part produced.” He then asks: “but whence the expenditure of energy causing that friction, and which must be itself accounted for? To answer this question he turns to the mechanical theory of heat and goes on to loosely outline how the heart is what he calls a “force-pump”, which receives blood and sends it to every part of the body, as discovered by William Harvey, that “acts like the piston of an engine and is dependent upon and consequently due to the cycle of nutrition and excretion which sustains physical or organic life.” It is likely, here, that McCulloch was modeling parts of this argument on that of the famous Carnot cycle. In conclusion, he summarizes his first and second law argument as such:
What is life?Later, building on this premise, in the famous 1944 book What is Life?, Nobel-laureate physicist Erwin Schrödinger theorizes that life, contrary to the general tendency dictated by the Second law of thermodynamics, decreases or maintains its entropy by feeding on negative entropy.[4] In a note to What is Life?, however, Schrödinger explains his usage of this term:
This is what is argued to differentiate life from other forms of matter organization. In this direction, although life's dynamics may be argued to go against the tendency of second law, which states that the entropy of an isolated system tends to increase, it does not in any way conflict or invalidate this law, because the principle that entropy can only increase or remain constant applies only to a closed system which is adiabatically isolated, meaning no heat can enter or leave. Whenever a system can exchange either heat or matter with its environment, an entropy decrease of that system is entirely compatible with the second law.[5] In 1964, James Lovelock was among a group of scientists who were requested by NASA to make a theoretical life detection system to look for life on Mars during the upcoming space mission. When thinking about this problem, Lovelock wondered “how can we be sure that Martian life, if any, will reveal itself to tests based on Earth’s lifestyle?” [6] To Lovelock, the basic question was “What is life, and how should it be recognized?” When speaking about this puzzling issue with some of his colleagues at the Jet Propulsion Laboratory, he was asked, well what would you do to look for life on Mars? To this Lovelock replied:
Thus, according to Lovelock, to find signs of life, one must look for a “reduction or a reversal of entropy.” Gibbs free energyIn recent years, the thermodynamic interpretation of evolution in relation to entropy has begun to utilize the concept of the Gibbs free energy, rather than entropy. This is because biological processes on earth take place at roughly constant temperature and pressure, a situation in which the Gibbs free energy is an especially useful way to express the second law of thermodynamics. The Gibbs free energy is given by: The minimization of the Gibbs free energy is a form of the principle of minimum energy, which follows from the entropy maximization principle for closed systems. Moreover, the Gibbs free energy equation, in modified form, can be utilized for open systems when chemical potential terms are included in the energy balance equation. In a popular 1982 textbook Principles of Biochemistry by noted American biochemist Albert Lehninger, it is argued that the order produced within cells as they grow and divide is more than compensated for by the disorder they create in their surroundings in the course of growth and division. In short, according to Lehninger, "living organisms preserve their internal order by taking from their surroundings free energy, in the form of nutrients or sunlight, and returning to their surroundings an equal amount of energy as heat and entropy."[7] Similarly, according to the chemist John Avery, from his recent 2003 book Information Theory and Evolution, we find a presentation in which the phenomenon of life, including its origin and evolution, as well as human cultural evolution, has its basis in the background of thermodynamics, statistical mechanics, and information theory. The (apparent) paradox between the second law of thermodynamics and the high degree of order and complexity produced by living systems, according to Avery, has its resolution "in the information content of the Gibbs free energy that enters the biosphere from outside sources."[8] References
Further reading
See also
|
||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Entropy_and_life". A list of authors is available in Wikipedia. |