To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Polydimethylsiloxane
Polydimethylsiloxane (PDMS) is the most widely used silicon-based organic polymer, and is particularly known for its unusual rheological (or flow) properties. Its applications range from contact lenses and medical devices to elastomers, caulking, lubricating oils and heat resistant tiles. PDMS is optically clear, and is generally considered to be inert, non-toxic and non-flammable. It has been assigned CAS number 63148-62-9, and is occasionally called dimethicone. It is one of several types of silicone oil (polymerized siloxane). Product highlight
ChemistryThe chemical formula for PDMS is (H3C)3[Si(CH3)2O]nSi(CH3)3, where n is the number of repeating monomer [SiO(CH3)2] units. Industrial synthesis can begin from dimethylchlorosilane and water by the following net reaction:
During polymerization, this reaction evolves potentially hazardous hydrogen chloride gas. For medical uses, a process was developed where the chlorine atoms in the silane precursor were replaced with acetate groups, so that the reaction product of the final curing process is nontoxic acetic acid (vinegar). As a side effect, the curing process is also much slower in this case. This is the chemistry used in consumer applications, such as silicone caulk and adhesives. Silane precursors with more acid-forming groups and fewer methyl groups, such as methyltrichlorosilane, can be used to introduce branches or cross-links in the polymer chain. Ideally, each molecule of such a compound becomes a branch point. This can be used to produce hard silicone resins. Similarly, precursors with three methyl groups can be used to limit molecular weight, since each such molecule has only one reactive site and so forms the end of a siloxane chain. The polymer is manufactured in multiple viscosities, ranging from a thin pourable liquid (when n is very low), to a thick rubbery semi-solid (when n is very high). PDMS molecules have quite flexible polymer backbones (or chains) due to their siloxane linkages, which are analogous to the ether linkages used to impart rubberiness to polyurethanes. Such flexible chains become loosely entangled when molecular weight is high, which results in PDMS having an unusually high level of viscoelasticity. Mechanical propertiesPDMS is viscoelastic, meaning that at long flow times (or high temperatures), it acts like a viscous liquid, similar to honey. However at short flow times (or low temperatures) it acts like an elastic solid, similar to rubber. In other words, if you leave some PDMS on a surface overnight (long flow time), it will flow to cover the surface and mold to any surface imperfections. However if you roll the same PDMS into a sphere and throw it onto the same surface (short flow time), it will bounce like a rubber ball.
Although the viscoelastic properties of PDMS can be intuitively observed using the simple experiment described above, they can be more accurately measured using dynamic mechanical analysis. This involves using a specialized instrument to determine the material's flow characteristics over a wide range of temperatures, flow rates, and deformations. Because of PDMS's chemical stability, it is often used as a calibration fluid for this type of experiment. The shear modulus of PDMS varies with preparation conditions, but is typically in the range of 100 kPa to 3 MPa. The loss tangent is very low ( Chemical compatibilityAfter polymerization and cross-linking, solid PDMS samples will present an external hydrophobic surface.[2] This surface chemistry makes it difficult for polar solvents (such as water) to wet the PDMS surface, and may lead to adsorption of hydrophobic contaminants. Plasma oxidation can be used to alter the surface chemistry, adding silanol (SiOH) groups to the surface. This treatment renders the PDMS surface hydrophilic, allowing water to wet (this is frequently required for, e.g. water-based microfluidics). The oxidized surface resists adsorption of hydrophobic and negatively charged species. The oxidized surface can be further functionalized by reaction with trichlorosilanes. Oxidized surfaces are stable for ~30 minutes in air, after a certain time hydrophobic recovery of the surface is inevitable independently of the surrounding medium whether it is vacuum, air or water.[3] Solid PDMS samples (whether surface oxidized or not) will not allow aqueous solvents to infiltrate and swell the material. Thus PDMS structures can be used in combination with water and alcohol solvents without material deformation. However most organic solvents will diffuse into the material and cause it to swell,[2] making them incompatible with PDMS devices. Despite this, some organic solvents lead to sufficiently small swelling that they can be used with PDMS, for instance within the channels of PDMS microfluidic devices. The swelling ratio is roughly inversely related to the solubility parameter of the solvent. Diisopropylamine swells PDMS to the greatest extent, solvents such as chloroform, ether, and THF swell the material to a large extent. Solvents such as acetone, 1-propanol, and pyridine swell the material to a small extent. Alcohols and polar solvents such as methanol, glycerol and water do not swell the material appreciably.[4] ApplicationsMany people are indirectly familiar with PDMS because it is an important (4%) component in Silly Putty, to which PDMS imparts its characteristic viscoelastic properties. The rubbery, vinegary-smelling silicone caulks, adhesives, and aquarium sealants are also well-known. PDMS is also used as a component in silicone grease and other silicone based lubricants, as well as in defoaming agents, mold release agents, damping fluids, heat transfer fluids, polishes, cosmetics, hair conditioners and other applications. PDMS has also been used as a filler fluid in breast implants, although this practice has decreased somewhat, due to safety concerns. It continues to be used in knuckle replacement implants, with good results. Activated dimethicone, a mixture of polydimethylsiloxanes and silicon dioxide (sometimes called simethicone), is used in Over-the-counter drug as an anti-foaming agent and carminative. As a food additive, it has the E number E900 and is used as an anti-foaming agent and an anti-caking agent. PDMS is commonly used as a stamp resin in the procedure of soft lithography, making it one of the most common materials used for flow delivery in microfluidics chips. PDMS can be cross-linked into networks and is a commonly used system for studying the elasticity of polymer networks. PDMS can be used in the treatment of head lice. Dimethicone is also used widely in skin moisturizing lotions, listed as an active ingredient whose purpose is "skin protectant." Some cosmetic formulations use dimethicone and related siloxane polymers in concentrations of use up to 15%. The Cosmetic Ingredient Review's (CIR) Expert Panel, has concluded that dimethicone and related polymers are "safe as used in cosmetic formulations" [1] PDMS is also used in analytical chemistry as a component of some types of SPME fibers. See also
References
Categories: Silicones | Cosmetic chemicals |
|||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Polydimethylsiloxane". A list of authors is available in Wikipedia. | |||||||||||||||||||||||||||||





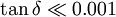 ).
).

