To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Crooks fluctuation theoremThe Crooks equation (CE) is an equation in Statistical Mechanics that relates the work done on a system during a non-equilibrium transformation to the free energy difference between the final and the initial state of the transformation. During the non equilibrium transformation the system is at constant volume and in contact with a heat reservoir. The CE is named after the chemist Gavin E. Crooks (then at University of California) who discovered it in 1998. The CE is a special case of the more general fluctuation theorem. Product highlightIf we define a generic reaction coordinate of the system as a function of the Cartesian coordinates of the constituent particles ( e.g. , a distance between two particles), we can characterize every point along the reaction coordinate path by a parameter λ, such that λ = 0 and λ = 1 correspond to two ensembles of microstates (see microstate (statistical mechanics)) for which the reaction coordinate is constrained to different values. A dynamical process where λ is externally driven from zero to one, according to an arbitrary time scheduling, will be referred as forward transformation , while the time reversal path will be indicated as backward transformation. Given these definitions, the CE sets a relation between the following four quantities:
The CE equation reads as follows:
In the
previous equation the difference WAB − ΔF
corresponds to the work dissipated in the forward transformation,
Wd. The probabilities
Note that for the backward transformation, the work distribution function must be evaluated by taking the work with the opposite sign. The two work distributions for the forward and backward processes cross at W = ΔF. This fact has been experimentally verified using optical tweezers for the process of unfolding and refolding of a small RNA hairpin and an RNA three-helix junction [1] The CE implies the Jarzynski equality. References
|
||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Crooks_fluctuation_theorem". A list of authors is available in Wikipedia. |





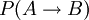 , i.e. the joint probability of taking a microstate
, i.e. the joint probability of taking a microstate 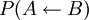 , i.e. the joint probability of taking the microstate
, i.e. the joint probability of taking the microstate ![\frac{P(A \rightarrow B)} {P( A \leftarrow B)} = \exp [ \beta ( W_{A \rightarrow B} - \Delta F )]](images/math/d/1/9/d196d9c9fdf9e510a1bd4c8b6b46abf9.png)
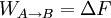 and
and 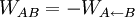 , and grouping together all the trajectories
yielding the same work (in the forward and backward transformation),
we can write the above equation in terms of the work distribution
functions as follows
, and grouping together all the trajectories
yielding the same work (in the forward and backward transformation),
we can write the above equation in terms of the work distribution
functions as follows
![P_{A \rightarrow B} (W) = P_{A \leftarrow B}(- W) ~ \exp[\beta (W - \Delta F)]](images/math/7/1/4/71449d90872d2ee3edbea697c40ddf76.png)


