Stabilizing sulfur cathode by single Li-ion channel polymer binder
Advertisement
The growing demands on the high-performance energy-storage system for emerging technologies such as electric vehicles and artificial intelligence drive the development of high-performance batteries. As a promising candidate of next-generation batteries, Li-S batteries have been drawn much attention carrying a high specific capacity (1675 mAh g-1) and energy density (2600 Wh kg-1). However, the diffusion of polysulfide in electrolyte cause changes in the structure of the sulfur cathode during discharge-charge cycles, which greatly limits the commercial applications of Li-S batteries.
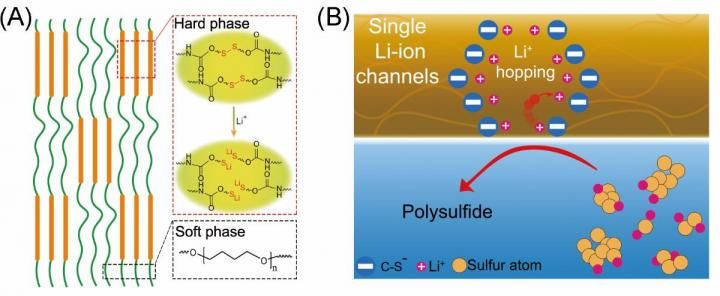
(A) Schematic diagrams of the formation of the polymer with the single-ion channels. (B) Schematic diagrams of single-ion channels regulate the polysulfide and Li-ion hopping.
©Science China Press
Polymer binder, as an essential component of electrode, acts to bond the active material and are related to the performance of batteries. Unfortunately, the conventional binder has failed to meet the requirements of emerging batteries. For example, the PVDF binder exhibits low ionic conductivity of Li-ions, poor mechanical stability, and almost none inhibition on the shuttle of polysulfide, these factors limit the applications of Li-S batteries. Therefore, an ideal polymer binder which overcomes the drawback of conventional binders is urgently needed for Li-S batteries.
In a new research published in the Beijing-based National Science Review, scientists at the Soochow Institute for Energy and Materials Innovations for Lithium-sulfur battery in Suzhou, China present the latest advances in Single Lithium-ion Channel Polymer Binder for Li-S battery. Co-authors Chaoqun Niu, Jie Liu, Xiaowei Shen, Jinqiu Zhou, Tao Qian and Chenglin Yan report a novel polymer binder with single lithium-ions channels allowing fast lithium-ions transport while blocking the shuttle of polysulfide anions. This study reports a new avenue to assemble a polymer binder with single lithium-ion channel for solving the serious problem of energy attenuation of Li-S batteries.
These scientists confirme the effect of the prepared polymer binder on Li-S batteries by monitoring polysulfide concentration in the electrolyte and device capacity retention in real time during the cycle. "The polymer binder is confirmed to effectively immobilize the shuttle effect of polysulfide intermediates by the in-situ UV-vis measurement." "Moreover, the excellent adhesion and mechanical stability of prepared binder maintain the structure integrity of sulfide cathode after discharge-charge cycles. These results demonstrate that the promising improvement of Li-S battery by the prepared binder and we believe the reported polymer binder with single Li-ion channels is one of the most effective strategies for the high-energy Li-S batteries."
Original publication
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic World Battery Technology
The topic world Battery Technology combines relevant knowledge in a unique way. Here you will find everything about suppliers and their products, webinars, white papers, catalogs and brochures.

Topic World Battery Technology
The topic world Battery Technology combines relevant knowledge in a unique way. Here you will find everything about suppliers and their products, webinars, white papers, catalogs and brochures.