The expanding possibilities of bio-based polymers
Researchers present a new route to prepare biobased polyesters with tuneable properties
Advertisement
Finding innovative and sustainable solutions to our material needs is one of the core objectives of green chemistry. The myriad plastics that envelop our daily life - from mattresses to food and cars - are mostly made from oil-based monomers which are the building blocks of polymers. Therefore, finding bio-based monomers for polymer synthesis is attractive to achieve more sustainable solutions in materials development.
ICIQ/Francesco Della Monica
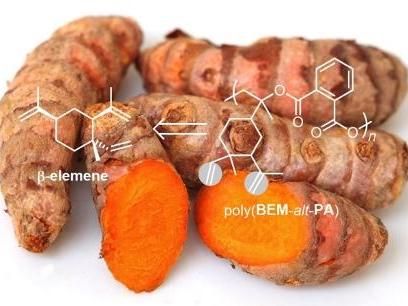
In a paper published in ACS Sustainable Chemistry & Engineering, researchers from the Kleij group present a new route to prepare biobased polyesters with tuneable properties. The researchers are building upon the multifunctional structure of the terpene β-elemene: three double bonds which have distinct reactivity, allowing to selectively transform these bonds and thus tweaking the functionalities in the backbone of the polymer. "This multi-functional terpene scaffold is rather unique and allows to fine-tune structural diversity and prospectively to modulate polymer and material properties," explains Arjan Kleij, ICIQ group leader and ICREA professor.
In collaboration with the company Isobionics, the researchers utilised β-elemene obtained through an innovative sugar-fermentation route. This process has proved to be a promising start for the use of β-elemene as a raw material for polymerisation. "Isobionic's sugar-fermentation route completely changes the scale of β-elemene availability, which now can be used in polymer production," explains Francesco Della Monica, postdoctoral researcher in the Kleij group working in the European SUPREME project, an MSCActions Individual Fellowship and first author of the paper.
Via a ring-opening copolymerisation reaction (ROCOP), the researchers combined β-elemene oxides and phthalic anhydride (a common monomer used in the preparation of polyesters) to create the biobased linear polymer poly(BEM-alt-PA) and its related structure, crosslinked-poly(BED-alt-PA). These transformations were achieved with catalytic systems (iron and aluminium aminotriphenolate complexes combined with bis-(triphenylphosphine) iminium chloride) developed previously by the group using non-critical, abundant elements for catalytic polymerisation.
Once the polyester is prepared, there are two remaining double bonds from the original terpene building block that can be easily and selectively addressed and functionalised, allowing to tailor the final polyester. "These post-modification reactions on a biobased polymer are quite rare. Most of the biobased monomers that are available don't present functionalities," remarks Della Monica.
The paper is a starting point for further development of β-elemene based polymers that allow tailoring the properties of the final material (depending on its use) through easy post-polymerisation modifications. The paper does not address the biodegradation of the material, although for Della Monica, "depending on the final use, the ideal thing may not be biodegradation but to create a recyclable polymer: i.e., take a starting material, create the polymer, use it, recover it, and then degrade it in a controlled fashion and reuse that material. Now that we have the idea of a circular economy within grasp, we need circular processes," concludes the scientist.
Original publication
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.

Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.