Method found for analysing complex, tiny crystals
Advertisement
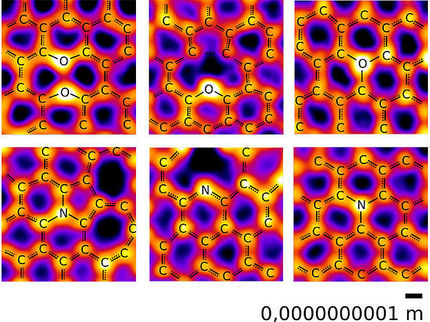
The atomic structure of solid substances can often be analysed quickly, easily and very precisely using X-rays. However, this requires that Crystals of the corresponding substances exist. Chemist Professor Oliver Oeckler from Leipzig University and his team are developing methods to make this possible even for very small crystals that cannot be seen with the naked eye. These include phosphorus oxide nitrides, which consist of phosphorus, nitrogen and oxygen and do not occur in nature. Unusual properties are attributed to this novel class of compounds, which have been difficult to access until now, because of their unusual structures.
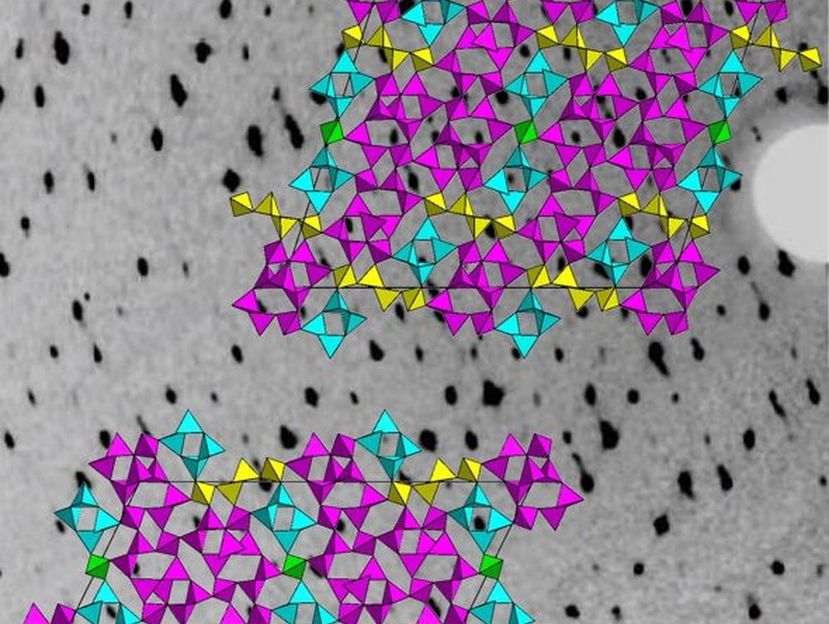
The image shows a section of the experimental data in the background, an X-ray diffraction pattern of the phosphorus oxide nitrides. In the foreground are sections of the crystal structure.
Universität Leipzig
Working with Professor Wolfgang Schnick from the Ludwig Maximilian University of Munich, Oeckler and his team have developed a method that made it possible, over a decade of research, to determine the complex crystal structure of new phosphorus oxide nitrides. The scientists have just published their findings in Chemistry – A European Journal.
When analysing crystal structure, the combination of electron microscopy and synchrotron radiation – particularly intense X-rays generated using a special technique at a large-scale research facility – plays a decisive role. However, the analysis of phosphorus oxide nitride shows that this is sometimes not enough. The substance, which could form the basis for novel phosphors in future studies, for example, was already produced for the first time in 2014, but its structure has not yet been elucidated, because it was previously considered a class of compounds that was difficult to access. Daniel Günther, a doctoral researcher in Oeckler’s working group, has now been able to solve the puzzle together with his mentor. “It was not due to the data, but to a trick of nature. We are not talking about just one substance, but three very complicated, intergrown compounds,” explains Günther, who is the first author of the study.
Sections of the atomic arrangements formed a kind of modular system from which complicated and also disordered structures can emerge. “Such an investigation requires extremely meticulous work, for which only a few staff members can muster the necessary patience and concentration. Without a research sabbatical and such a dedicated member of staff, it probably wouldn’t have worked. Most people would have been horrified by what at first glance appeared to be ‘unanalysable’ data and would never have mentioned it again,” says Oliver Oeckler. He points out that the significant factor here is not only the structure of oxonitridophosphates, which the researchers find very interesting, but also the method of analysis. The procedure described in their article could be used to solve similar analytical problems with completely different substances.