Materials science meets medicine: Why implants fail
Advertisement
What happens to titanium implants in the body? Why are they sometimes rejected or even break? Empa researcher Martina Cihova is looking for answers to these questions at the interface between the implant and the body, between materials science and medicine. She recently received an "Ambizione Grant" from the Swiss National Science Foundation (SNSF) for her research project.

Empa researcher Martina Cihova is investigating how the body can attack the surface of titanium implants.
Empa

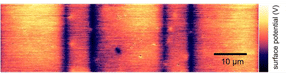
The image from the atomic force microscope shows clearly recognizable areas with different surface potentials, which were created by laser-induced changes in the structure of the titanium oxide.
Empa

Thanks to medical advances, we humans are living longer and longer. Understandably, we want to stay healthy and mobile into old age. Implants and prostheses replace "worn out" joints and teeth, stop pain and improve quality of life. Modern medical implants are small marvels of biomaterial and bioengineering at the same time. Nevertheless, implants occasionally fail, which can have serious consequences for patients.
Why do these failures occur - and why have they increased rather than decreased in recent years? Empa researcher Martina Cihova from the "Joining Technology and Corrosion" laboratory wants to find answers to these questions. To do so, she is taking a close look at the behavior of implants in the body - or, to be more precise, under the microscope. The scientist has been awarded a four-year "Ambizione Grant" from the Swiss National Science Foundation for her research project.
Many implants - including artificial joints, dental implants and pacemakers - are made of titanium. This transition metal is light and stable, is very durable in the body and allows bone to grow particularly well. These properties are due to a thin oxide layer that forms on the surface of the titanium when it comes into contact with air. Ultimately, it is not the titanium itself, but the protective layer on the surface of the implants that comes into contact with the body. "As this natural passive layer is less than ten nanometres thick, it is often given too little attention in medical technology and research," says Martina Cihova.
In addition, some manufacturers modify the oxide layer, for example by thickening it to give different implant sizes or models a color coding and thus make work easier for doctors. Others roughen the surface of the implants so that the bone can grow better - or engrave the serial number with a laser. 3D printing of patient-specific implants is also possible today using laser processes. These are all sensible applications, but: "Any surface treatment can change the titanium oxides on the surface," says Cihova, "and too little research has been done into what this means for the interaction of the implant with the body and for its corrosion resistance."
Research at the frontier
The Empa researcher wants to close this knowledge gap with her project. Even as a bioengineering student, Cihova was enthusiastic about materials science. She therefore embarked on a new path for her doctorate - metallurgy - to further deepen her interest in materials. Now she is combining her expertise in the two fields and focusing it precisely on the point where metal, or rather metal oxides, and biology meet: the interface between the implant and the human body.
"Such biointerfaces are highly complex, but also extremely interesting," says the young researcher. "When you think of corrosion, you think of salty seawater, humid air, maybe a rusty bicycle - but not the human body." Yet the human body can be a surprisingly aggressive environment, especially when immune reactions take place. Immune cells release various substances that can, among other things, lower the pH value and attack the implant. So what does the body do with materials that we consider stable? This is precisely where research into biocorrosion comes in.
These processes are very complex on an (electro)chemical and biological level. In addition, not all titanium oxide is the same. It can take on three different crystalline forms - all with the same chemical composition, TiO2 - or be amorphous, structurally "undefined", so to speak. All these forms differ in their electronic and electrochemical properties and therefore potentially also in their interactions with the body.
Increasing complexity in a controlled manner
The surface treatment of implants can change the crystal forms of the oxides, either on the entire implant or only selectively. In order to understand the effects of this local heterogeneity in particular on the already complex biointerface, a structured approach is required. First, Cihova and her team, in collaboration with the Empa experts for laser processing of metals in Thun, produce sample substrates with differently structured titanium oxide layers that vary systematically in their heterogeneity. These substrates are then successively exposed to increasingly complex body fluids in order to investigate the fundamental relationships between the structure, properties and reactivity of the oxides.
"We start with simulated physiological fluids that only contain water and ions," explains Cihova. The next step is to add proteins, such as fibrinogen, which is involved in the immune response and wound healing. Finally, the researchers plan to investigate how the biointerface behaves in contact with living macrophage cells - the "police of the body". To this end, they are collaborating with Empa researchers in St. Gallen. "I am very pleased that we were able to inspire colleagues from all three Empa sites for this project," says Cihova. "This enables us to tackle such complex issues in an interdisciplinary way."
In each of these steps, the interfaces are examined "through their paces" using electrochemical methods paired with high-resolution electron and atomic force microscopy. "Seeing is understanding - even if that means looking at a size scale that is far smaller than a human cell," says Cihova. "This is where crucial details can often be discovered."
The Empa researcher hopes that the findings from the next few years will lead to safer and more stable implants. And also "that we learn more about how the fascinating range of oxide properties can be used specifically in biomedicine." Following her "Ambizione" project in 2028, she also wants to extend the new methods to other medical materials. Cihova is convinced that this field of research will become even more important in the future: "The behavior of metal oxides at biointerfaces is also key to their performance in the emerging fields of nanomedicine and implantable sensor technology."
Note: This article has been translated using a computer system without human intervention. LUMITOS offers these automatic translations to present a wider range of current news. Since this article has been translated with automatic translation, it is possible that it contains errors in vocabulary, syntax or grammar. The original article in German can be found here.































































