The Schrödinger equation as a Quantum clock
Researchers succeed in controlling multiple quantum interactions in a realistic material
Materials with controllable quantum mechanical properties are of great importance for the electronics and quantum computers of the future. However, finding or designing realistic materials that actually have these effects is a big challenge. Now, an international theory and computational team led by Cesare Franchini from the University of Vienna, find that multiple quantum interactions can coexist in a single real material and show how an electric field can be used to control them.
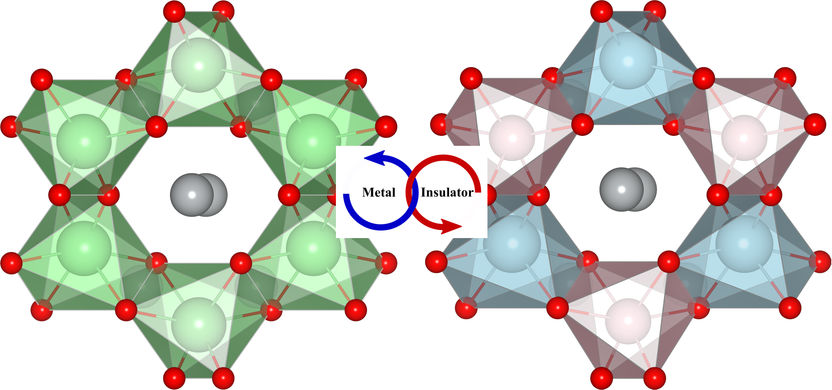
The application of an electric field changes the symmetry of the crystal and drives a transition from a metal (left) to an insulator (right).
Copyright He/Franchini
Next generation electronics and quantum computers rely on materials that exhibit quantum-mechanical phenomena and related properties, which can be controlled by external stimuli, e.g. by a battery in a microelectronic circuit. Quantum mechanics governs, for example, how fast – and if at all– electrons can move through a material and, thereby, determine whether the material is a metal which conducts an electric current or whether it is an insulator which cannot conduct a current. Furthermore, the interaction of the electrons with the crystal structure controls whether a material can be ferroelectric. In this case one can switch between two electric orientations by applying an external electric field. The possibility to activate multiple quantum-mechanical properties in one single material is of fundamental scientific interest but can also expand the spectrum of potential applications.
An international team of researchers led by Professor Cesare Franchini and Dr. Jiangang He of the Quantum Materials Modelling Group at the University of Vienna, in cooperation with Professor Rondinelli of Northwestern University and Professor Xing-Qiu Chen of the Chinese Academy of Science has now demonstrated that multiple quantum interactions can, indeed, coexist in a single material and that it is possible to tune between them with an electric field. “This is like awakening different kinds of quantum interactions that are quietly sleeping in the same house without knowing each other”, explains Prof. Franchini. For their discovery the scientists solved the relativistc form of the Schrödinger equation, by performing computer simulations on the Vienna Scientific Cluster. The material of their choice, the compound Ag2BiO3, is exceptional for two reasons; on the one hand it is composed of the heavy element bismuth, which allows the spin of the electron to interact with its own motion (spin-orbit coupling) - a feature that has no analogy in classical physics. On the other hand, its crystal structure does not exhibit inversion symmetry, suggesting that ferroelectricity could occur.
“Harmonizing multiple quantum mechanical properties which often do not coexist together and trying to do it by design is highly complex,” states Professor Rondinelli.The application of an electric field to the oxide Ag2BiO3 changes the atomic positions and determines whether the spins are coupled in pairs (forming so-called Weyl-fermions) or separated (Rashba-splitting), and whether the material is electrically conductive or not. “We have found the first real case of a topological quantum transition from a ferroelectric insulator to a non-ferroelectric semi-metal”, beams Prof. Franchini. The spin-orbit coupling is of fundamental importance, as it can yield the formation of novel quantum states of matter, and represents one of the hottest research area in modern physics. Also in view of potential applications, there are promising prospects: the control over quantum interactions in a real material could enable ultrafast, low-power electronics and quantum computers for qualitative leaps forward in data acquisition, processing, and exchange.
Original publication
Other news from the department science

Get the chemical industry in your inbox
From now on, don't miss a thing: Our newsletter for the chemical industry, analytics, lab technology and process engineering brings you up to date every Tuesday and Thursday. The latest industry news, product highlights and innovations - compact and easy to understand in your inbox. Researched by us so you don't have to.


























































