To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
BaroclinityIn fluid dynamics, the baroclinity (sometimes called baroclinicity) is a measure of the stratification in a fluid. A baroclinic atmosphere is one for which the density depends on both the temperature and the pressure; contrast this with barotropic atmosphere, for which the density depends only on the pressure. In atmospheric terms, the barotropic zones of the Earth are generally found in the central lattitudes, or tropics, whereas the baroclinic areas are generally found in the mid-lattitude/polar regions. Product highlightBaroclinity is proportional to which again is proportional to the angle between surfaces of constant pressure and surfaces of constant density. Thus, in a barotropic fluid (which is defined by zero baroclinity), these surfaces are parallel. Areas of high atmospheric baroclinity are characterized by the frequent formation of cyclones. Baroclinic instabilityBaroclinic instability is a fluid dynamical instability of fundamental importance in the atmosphere and in the oceans. In the atmosphere it is the dominant mechanism shaping the cyclones and anticyclones that dominate weather in mid-latitudes. In the ocean it generates a field of mesoscale (100 km or smaller) eddies that play various roles in oceanic dynamics and the transport of tracers. Baroclinic instability is a concept relevant to rapidly rotating, strongly stratified fluids. Whether one is rapidly rotating or not is determined in this context by the Rossby number, which is a measure of how close the flow is to solid body rotation. More precisely, a flow in solid body rotation has vorticity that is proportional to its angular velocity. The Rossby number is a measure of the departure of the vorticity from that of solid body rotation. The Rossby number must be small for the concept of baroclinic instability to be relevant. When the Rossby number is large, other kinds of instabilities, often referred to as inertial, becomes more relevant. The simplest example of a stably stratified flow is an incompressible flow with density decreasing with height. In a compressible gas such as the atmosphere, the relevant measure is the vertical gradient of the entropy, which must increase with height for the flow to be stably stratified. One measures the strength of the stratification by asking how large the vertical shear of the horizontal winds has to be in order to destabilize the flow and produce the classic Kelvin-Helmholtz instability. This measure is the Richardson number. When the Richardson number is large, the stratification is strong enough to prevent this shear instability. Before the classic work of Jule Charney and Eric Eady on baroclinic instability in the late 1940's, most theories trying to explain the structure of mid-latitude eddies took as their starting points the high Rossby number or small Richardson number instabilities familiar to fluid dynamicists at that time. The most important feature of baroclinic instability is that it exists even in the situation of rapid rotation (small Rossby number) and strong stable stratification (large Richardson's number) typically observed in the atmosphere. The energy source for baroclinic instability is the potential energy in the environmental flow. As the instability grows, the center of mass of the fluid is lowered. In growing waves in the atmosphere, cold air moving downwards and equatorwards displaces the warmer air moving polewards and upwards. Baroclinic instability can be investigated in the laboratory using a rotating, fluid filled annulus. The annulus is heated at the outer wall and cooled at the inner wall, and the resulting fluid flows give rise to baroclinically unstable waves. The term "baroclinic" refers to the mechanism by which vorticity is generated. Vorticity is the curl of the velocity field. in general, the evolution of vorticity can be broken into contributions from advection (as vortex tubes move with the flow), stretching and twisting (as vortex tubes are pulled or twisted by the flow) and baroclinic vorticity generation, which occurs whenever there is a density gradient along surfaces of constant pressure. Baroclinic flows can be contrasted with barotropic flows in which density and pressure surfaces coincide and there is no baroclinic generation of vorticity. The study of the evolution of these baroclinic instabilities as they grow and then decay is synonymous in dynamical meteorology with the problem of developing theories for the fundamental characteristics of midlatitude weather. Baroclinic vectorBeginning with the equation of motion for a fluid (say, the Euler equations or the Navier-Stokes equations) and taking the curl, one arrives at the equation of motion for the curl of the fluid velocity, that is to say, the vorticity. In a fluid that is not all of the same density, a source term appears in the vorticity equation whenever surfaces of constant density (isopycnic surfaces) and surfaces
of constant pressure (isobaric surfaces) are not aligned. This term (denoted
by the subscript
This vector is of interest both in compressible fluids and in incompressible (but inhomogenous) fluids. Internal gravity waves as well as unstable Rayleigh-Taylor modes can be analyzed from the perspective of the baroclinic vector. It is also of interest in the creation of vorticity by the passage of shocks through inhomogenous media, such as in the Richtmeyer-Meshkov instability. Divers may be familiar with the very slow waves that can be excited at a thermocline or a halocline; these are internal waves. Similar waves can be generated between a layer of water and a layer of oil. When the interface between these two surfaces is not horizontal and the system is close to hydrostatic equilibrium, the gradient of the pressure is vertical but the gradient of the density is not. Therefore the baroclinic vector is nonzero, and the sense of the baroclinic vector is to create vorticity to make the interface level out. In the process, the interface overshoots, and the result is an oscillation which is an internal gravity wave. Unlike surface gravity waves, internal gravity waves do not require a sharp interface. For example, in bodies of water, a gradual gradient in temperature or salinity is sufficient to support internal gravity waves driven by the baroclinic vector.
|
||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Baroclinity". A list of authors is available in Wikipedia. |





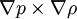
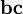 below) is known as the baroclinic vector. (Note that
below) is known as the baroclinic vector. (Note that
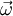 is the vorticity vector,
is the vorticity vector,