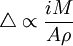To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
CoulometryCoulometry is the name given to a group of electroanalytical chemistry techniques that determine the amount of matter transformed during an electrolysis reaction by measuring the amount of electricity (in coulombs) consumed or produced. The technique is applicable to redox reactions, which are reactions in which electrons are transferred from one molecule to another. The reaction is controlled by applying an electrical potential and the amount of electricity (i.e, the number of electrons) needed to complete the reaction is the main measurement. There are two basic categories of coulometric techniques. One category, potentiostatic coulometry, involves holding the electric potential constant during the reaction using a potentiostat. The other, called coulometric titration or amperostatic coulometry, keeps the current (measured in amperes) constant using an amperostat. Both techniques take place within an electrochemical cell and are explained in more detail below. Coulometric techniques are as accurate as other analytical techniques used to carry out similar analyses, but are usually quicker and more convenient. Since coulometric methods do not measure directly the amount of matter transformed in the redox reaction, it is important that the reaction maintains a stoichiometrical relationship. Thus, each electron introduced or removed from the electrochemical cell must correspond to a known number of molecules in the reaction. When this is the case, standardization or calibration curves are not required for accurate analysis because the amount of current can be determined using the equation: Q = It where Q is the amount of electricity in coulombs, I is the current in amperes and t is the time in seconds. Q is then related to the reaction by the stoichiometric ratio. As the reaction progresses, the concentration of the molecule being converted will decrease. The electrical potential must be increased to maintain the direction of the reaction as the concentrations in the sample change. Eventually the electrical potential will be large enough that the solvent (usually water) begins to decompose. As a result, the system will have an excess of ions and will give an error in the calculation of the concentration of the reaction components, indicating that there are more molecules in the system than are actually present. To compensate for this error, an ion such as cerium (Ce3+) that is oxidized at a lower potential than water, is added to maintain the efficiency of the system. Product highlight
Potentiostatic coulometryPotentiostatic coulometry involves keeping the electrochemical potential at the working electrode (the electrode involved in the reaction) constant, which results in an exponential decrease in current as the reaction proceeds. This is because the potential at the working electrode is not equal to the potential of the entire cell. It is related by the Nernst equation:
where R is the gas constant, n is the stoichiometric number of electrons, and F is the Faraday constant. E0 is the cell potential and Q is the instantaneous ratio of the concentrations of the component in the system. This relationship explains the exponential decrease in current observed in a system at constant potential. Maintaining the constant potential at the working electrode allows for the primary component to react without the involvement of other components in the sample. An advantage to this kind of analysis over electrogravimetry is that it does not require that the product of the reaction be weighed. This is useful for reactions where the product does not deposit as a solid, such as the determination of the amount of arsenic in a sample from the electrolysis of arsenous acid (H3AsO3) to arsenic acid (H3AsO4). For situations like this, a reference electrode with a known and constant potential is added to the apparatus to measure the potential difference, so that changes to the current can be made qualitatively. This can also be used to determine the end of the reaction, which is when the current begins to approach zero. A potentiostat is a device that keeps the potential at the working electrode constant relative to the reference electrode. The circuit for the potentiostats works to keep the voltage constant by noticing changes in the resistance of the system and compensating inversely with a change in the current. As a result, a change to a higher resistance would cause the current to decrease to keep the voltage constant in the system. The cathode of a potentiostatic coulometry experiment is usually a platinum electrode with a large surface area or a mercury pool, although other particularly reactive electrodes can be used in rare circumstances. Coulometric titrationCoulometric titrations use a constant current system to perform the reaction. The only measurements required in these systems is the time it takes to complete the electrolysis. The product of this time and the current is then used to determine the total amount of electricity used. The endpoint of the titration can be determined analytically by using an indicator that is placed in the sample and signals when the system reaches equilibrium. Alternatively, the endpoint can be determined from data provided by potentiometric, amperometric or conductance measurements. This is similar to regular chemical titrations. Since concentration polarization is inevitable in coulometric titrations, most (if not all) of the reaction must occur distant from the working electrode. Otherwise, the potential will need to constantly increase as the reaction progresses to maintain the production of products. Therefore, coulometric titration reactions usually have other ions in the sample that react directly with the electrode and then react with the sample molecules. Coulometric titrations can also be done by back-titration. An amperostat is used to maintain a constant current in coulometric titrations. It reacts to changes in the resistance of the cell by altering its output potential. Both amperostats and potentiostats can be built relatively cheaply using operational amplifiers. ApplicationsPotentiostatic coulometry techniques have been used to determine over 50 elements. They are used very frequently for the determination of uranium and plutonium since potentiostatic coulometry techniques do not interfere much with the sample. Another useful application is the determination of the amount of oxygen in a sample by using a cadmium electrode and a porous silver electrode. This system for determining oxygen is convenient because it does not require an external power supply or a potentiostat to control the reaction since it does not reach a potential that is high enough to oxidize other substances. In addition, potentiostatic techniques can be used to separate materials, to measure the diffusion current, and to determine and sometimes synthesize organic compounds. Coulometric titrations can be used for the neutralization of acids, which contain H+ ions, by producing hydroxide ions (OH-) at an electrode to form water. Mercury, silver chloride, or silver bromide electrodes are good counter electrodes for this reaction. The coulometric technique has one real advantage over the volumetric technique, which is that the interference of carbonate ions (CO32-) is easily removed by bubbling a carbon-dioxide deficient gas through the sample. This removes carbon dioxide in the sample, which would otherwise form carbonate ions and interfere with the analysis. Other applications include:
Karl Fischer reactionThe Karl Fischer reaction uses coulometric titration to determine the amount of water in a sample. It can determine concentrations of water on the order of mg/L. It is applied to find the amount of water in a myriad of substances such as butter, sugar, cheese, paper, and petroleum. The working electrode for this analysis reaction is an iodine electrode, while the reference electrode is a platinum electrode. The reaction involves converting solid iodine into hydrogen iodide in the presence of sulfur dioxide and water. Methanol is most often used as the solvent, but monoether glycol and diethylene glycol also work. Pyridine, which is a base, is often used to counteract the formation of sulfuric acid, although the use of imidazole and diethanolamine for this role are becoming more common. All reagents must be anhydrous for the analysis to be quantitative. The balanced chemical equation, using methanol and pyridine, is: In this reaction, a single molecule of water reacts with a molecule of iodine. Since this technique is used to determine the water content of samples, atmospheric humidity could alter the results. Therefore, the system is usually isolated with drying tubes or placed in an inert gas container. In addition, the solvent will undoubtedly have some water in it so the solvent’s water content must be measured to compensate for this inaccuracy. To determine the amount of water in the sample, analysis must first be performed using either back or direct titration. In the direct method, just enough of the reagents will be added to completely use up all of the water. At this point in the titration, the current approaches zero. It is then possible to relate the amount of reagents used to the amount of water in the system via stoichiometry. The back-titration method is similar, but involves the addition of an excess of the reagent. This excess is then consumed by adding a known amount of a standard solution with known water content. The result reflects the water content of the sample and the standard solution. Since the amount of water in the standard solution is known, the difference reflects the water content of the sample. Determination of Film ThicknessA slightly different application of coulometry is the determination of the thickness of metallic coatings. This is performed by measuring the quantity of electricity needed to dissolve a well-defined area of the coating. The film thickness Δ is proportional to the constant current i, the molecular weight M of the metal, the density ρ of the metal, and the surface area A:
If the type of metal used for the coating is known then the amount of current necessary to remove the coating is proportional to the thickness of the coating. The electrodes for this reaction are a platinum electrode and an electrode that relates to the reaction. For tin coating on a copper wire, a tin electrode is used, while a sodium chloride-zinc sulfate electrode would be used to determine the zinc film on a piece of steel. Special cells have been created to adhere to the surface of the metal to measure its thickness. These are basically columns with the internal electrodes with magnets or weights to attach to the surface. The results obtained by this coulometric method are similar to those achieved by other chemical and metallurgic techniques. CoulometersElectronic coulometer
Electrochemical coulometers (synonym: voltameters)There are two common types of coulometers based on electrochemical processes:
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Coulometry". A list of authors is available in Wikipedia. |





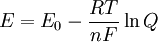
![\mathrm{[C_6H_5NH]SO_3CH_3 + I_2 + H_2O + 2 C_6H_5N} \longrightarrow \mathrm{[C_6H_5NH]SO_4CH_3 + 2 [C_6H_5NH]I}](images/math/3/d/a/3da9345a91f86fb0288e8080d18df76c.png)