Enzyme-metal-single-atom hybrid catalysts for efficient one-pot chemoenzymatic reactions
A promising strategy for effectively merging the enzymatic and single-atom catalysis
Advertisement
The merging of different types of catalysis including enzymatic, homogeneous, and heterogeneous catalysis is fundamentally important for both understanding catalysis at the atom level and the design of novel hybrid catalysts. The latter points to a direction toward the ideal catalyst that can drive complex tandem reactions efficiently in one-pot manner, and simplify the whole chemical production and separation process. Artificial enzymes that merge enzymatic, homogeneous, and heterogeneous catalysis provide such a promising platform for developing novel hybrid catalysts that can operate to achieve the above goal efficiently under ambient conditions.
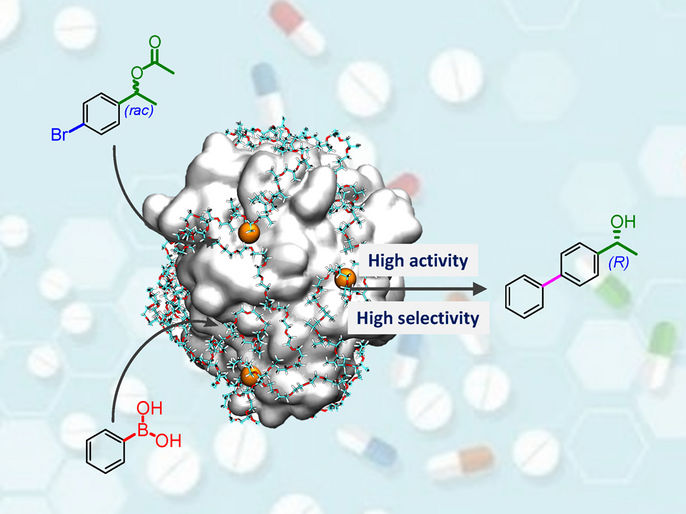
An one-pot chemoenzymatic cascade reaction to synthesize chiral alcohol was realized by using the highly active and selective enzyme-metal-single-atom hybrid as the catalyst. The enzyme-metal-single-atom hybrid catalyst provides a promising strategy for effectively merging the enzymatic and single-atom catalysis.
Chinese Journal of Catalysis
Directed evolution has been applied to tailor the active site of an enzyme with high efficiency, and artificial metalloenzymes have been generated by replacing the original metal atom at the active site by a new one. These methods are successful but usually only engineer the original active sites of enzymes, and this implies that the engineered enzyme may still be limited to catalyzing a single reaction. Inspired by the atomically dispersed metal catalysts that make a bridge between the homogeneous and heterogeneous catalysis, it is proposed that anchoring the metal single atoms to the nonactive sites of an enzyme can lead to an artificial enzyme with both its original active site and the new active sites of metal single atoms, merging the enzymatic and single-atom catalysis.
Recently, a research team led by Prof. Jun Ge from Tsinghua University reported the first enzyme-metal-single-atom hybrid catalyst that constructed by a photochemical approach. The Pd single atoms anchored lipase (Pd1/CALB-P) shows excellent activity and selectivity in the one-pot chemoenzymatic cascade reaction to asymmetric synthesize (R)-1-(4-biphenyl) ethanol. It can efficiently drive one-pot cascade reactions in aqueous solution at 30 oC to achieve facile synthesis of chiral biaryl alcohols, which are important pharmaceutical intermediates that traditionally require complex synthesis procedures. The rate of (R)-1-(4-biphenyl) ethanol formation catalyzed by Pd1/CALB-P is more than 30-fold higher than that of the combination of commercial palladium on carbon (Pd/C) and lipase-pluronic conjugate (CALB-P). The enzyme-metal-single-atom hybrid catalyst provides a promising strategy for effectively merging the enzymatic and single-atom catalysis.
Original publication
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.

Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.