New types of structures for cage-like clathrates
Advertisement
Cage-like compounds called clathrates could be used for harvesting waste heat and turning it into electricity. UC Davis chemists just discovered a whole new class of clathrates, potentially opening new ways to make and apply these materials.
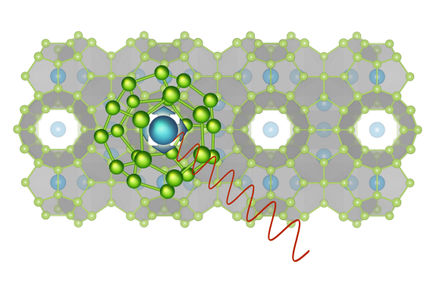
A clathrate is basically a cage of atoms with another atom trapped inside, said Kirill Kovnir, assistant professor of chemistry at UC Davis, who led the work, published recently in the journal Angewandte Chemie. Because the cage is relatively large compared to the atom, the trapped atom can rattle around inside, and that means that clathrates conduct heat very poorly, he said.
What they can do, though, is convert heat into electricity .
"Our energy sources waste about 60 percent or more as heat," Kovnir said. For example, a car engine generates lots of heat, almost none of which is usefully captured.
Thermoelectric devices that can convert heat to electricity have been used for example to power Mars rovers, where a radioactive source gives off heat that is converted to electricity to power the rover. Widely available thermoelectrics could be used for applications from powering a watch with body heat to making vehicles more efficient.
Clathrates of Metals and Other Atoms
Clathrates have been around for a long time -- as a class of materials, they were discovered in 1810 by the chemist Humphrey Davy. Clathrate structures based on water under pressure trap reserves of methane in the deep ocean.
Kovnir, however, is more interested in clathrates built of atoms such as copper, zinc and phosphorous and barium that are stable at room temperatures.
To date, all clathrates described have been based on a tetrahedral structure: each atom in the cage is bonded with four other atoms. More than 200 years after they were discovered, Kovnir's team has produced and described stable clathrates with atoms with five, six or more bonds.
"It was assumed that that framework had to be tetrahedrally coordinated," Kovnir said. "This is the first case where they don't have to be, and it suggests many more are possible."
The chemists were actually trying to probe the stability of the clathrate structure when they discovered the new compounds. To make four bonds, each atom needs four electrons available. By adding atoms with more electrons (such as zinc) Kovnir was expecting to be able to break the clathrate structure. Instead, they found they could produce entirely new, stable structures, including one with a cage of zinc, copper and phosphorous atoms trapping a barium atom. The new structure was featured on the cover of the journal Angewandte Chemie, with an accompanying research highlight.
Next steps are to optimize the thermoelectric properties of the new materials and see if they can tweak them for best performance, Kovnir said.