Creating chiral carbons
Advertisement
South Korean scientists have developed a racemisation-resistant substrate that can be selectively alkylated to make new chiral carbon centres.
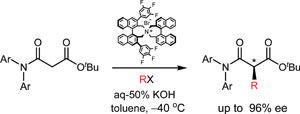
Mono-alkylation of a malonamic ester.
Although there are many ways to make chiral carbon centres by alkylating carbonyl compounds, until now scientists have been unable to asymmetrically mono-alkylate at the 2-position of 1,3-dicarbonyls because the compounds racemise easily under basic or acidic conditions.
Hyeung-geun Park, at Seoul National University, and colleagues converted one of the two ester groups on malonyl esters to an amide to reduce the acidity of the hydrogens at the 2-position. He showed that the resulting malonamic esters could be mono-alkylated under basic conditions at the 2-position with high enantioselectivity, indicating that the malonamic esters are racemisation-resistant.
'To the best of our knowledge, this is the first report to accomplish direct mono-alkylation of the 1,3-dicarbonyl system,' says Park. As well as reducing the acidity of the hydrogens, Park says he may have inhibited enolisation of the products by introducing strain between the N-substituent on the amide group and the new 2-substituent.
Chiral mono-alkylated malonyl derivatives are useful synthetic intermediates, explains Park, because they can be converted into diverse chiral building blocks. He has already selectively reduced the malonamic esters to give a range of products and plans to investigate more chemoselective transformations in the future.
Original publications: Mi-hyun Kim et al, Chem. Commun., 2009.