Research team synthesizes monometallic endohedral azafullerene
“This result is very interesting and helpful for understanding the unique properties of azafullerenes”
Advertisement
Recently, a research team led by Prof. YANG Shangfeng from the University of Sciences and Technology of China (USTC) of the Chinese Academy of Sciences (CAS) synthesized La@C81N, a type of monometallic endohedral azafullerene (MEAF) for the first time, providing a new way to modulate the electronic properties of metallic endohedral fullerenes through skeletal modifications of the carbon cage. Their work was published in Journal of the American Chemical Society.
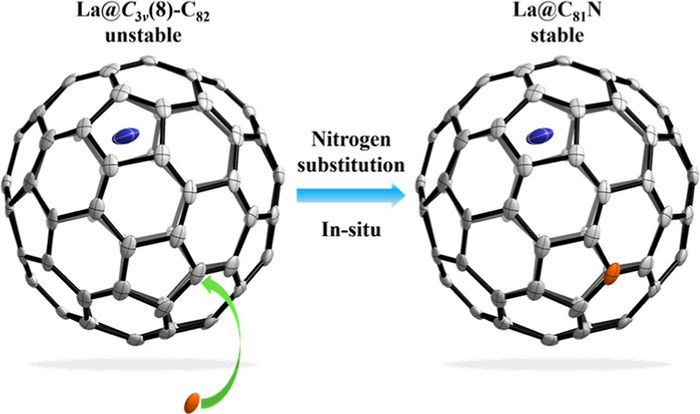
Nitrogen substitution of carbon atoms on La@C3v(8)-C82 carbon cage.
Image by XIANG Wenhao et al.
Azafullerenes are created when the carbon atoms on the cage are replaced by nitrogen atoms. Azafullerenes vary from fullerenes in both their electronic structures and physicochemical properties. Besides skeletal and exohedral modifications, fullerenes can also be modified endohedrally via encapsulating metal or non-metal atoms, exhibiting unique properties such as single-molecule magnetism. Therefore, to combine the advantages of both types of fullerenes, researchers have been attempting to modify the cage of endohedral metallofullerenes to obtain MEAF. Although previous research has detected ionized MEAF, [La@C81N]+, in gas phase, pristine MEAF has never been synthesized.
Based on previous works on the synthesis and isolation of novel low-stability heterofullerenes, Prof. YANG’s team synthesized pristine MEAF La@C81N by a modified Krätschmer-Huffman DC-arc discharge method and isolated La@C81N by a four-step high performance liquid chromatography (HPLC). To verify the carbon cage is an azafullerene, they characterized the molecular structure of La@C81N by single-crystal X-ray diffraction and confirmed that the carbon cage was derived from C3v(8)-C82 with one carbon atom replaced by a nitrogen atom.
Furthermore, the team systematically studied the electronic properties of La@C81N by ESR spectroscopy, UV-vis-NIR spectroscopy and electrochemistry. They also performed DFT calculations to identify the possible nitrogen substitution site. The results showed that La@C81N exists in a stable monomer form with a closed-shell electronic state, drastically different from the open-shell electronic state of La@C3v(8)-C82. T Therefore, skeletal modification of the fullerene carbon cage could regulate the electronic properties of metallic endohedral fullerenes.
This work tackled the two-decade challenge of synthesizing and isolating pristine MEAF. As commented by the reviewers, “this result is very interesting and helpful for understanding the unique properties of azafullerenes.”