Two Worlds, One Material
Polymers with a metal backbone
Advertisement
Until now, it has been clear: you can have a metal or a plastic, but not both in one. However, things don’t have to stay that way. In the journal Angewandte Chemie, a Chinese research team has now reported a polymer with a metallic backbone that is conductive, thermally stable, and has interesting optoelectronic properties.
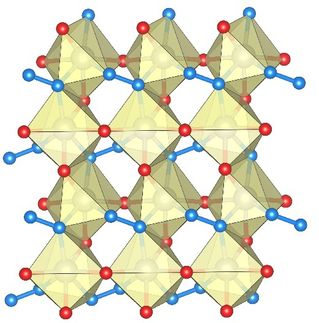
© Wiley-VCH
Because of the different electronic structures of metal and nonmetal atoms, it is difficult to confer the properties of metals, such as high thermal and electrical conductivity, upon polymers. Polymers with a metal backbone could combine the advantages of both types of material and open routes to materials with novel functionality. The problem lies in the weak bonds between the metal atoms, which cannot stabilize a polymer backbone to the same extent as the nonmetal atoms of conventional polymers. A team led by Guowei Wang and Huisheng Peng has now successfully synthesized a stable polymer with a backbone made of nickel atoms.
The team at Fudan University and East China University of Science and Technology (Shanghai, China) used a chalice-shaped molecule (calixarene) with four binding sites as the “scaffold” for the metal polymer. They attached four poly(aminopyridine) chains to the calixarenes, which bundles the four chains and aligns them in parallel. Synthesis of the chains can be carried out stepwise from individual building blocks or several larger blocks can be linked together. By using an iterative synthetic procedure in which protective groups and end caps are bound and then removed, the team is able to produce chains of equal length. They then carry out the metalation. The nitrogen atoms of the chain molecules can bind nickel. Their distance from each other exactly matches the distance of metal–metal bonds, causing the nickel atoms to link together in a single line. The four poly(aminopyridine) chains wrap around the nickel chain in a helix, as shown by X-ray structure analysis, and stabilize it. The team was thus able to synthesize polymers with a nickel backbone and precisely controlled length. They produced versions with three to 21 nickel atoms. Interestingly, the distance between nickel atoms decreases as the chain length increases, strengthening the Ni–Ni bonds.
The new materials may conduct electricity, are thermally stable, and can be processed in solution. They demonstrate strong length-dependent light absorption with narrow band gaps, which is promising for optoelectronic devices and semiconductors. The new synthetic strategy could also be expanded to other transition metals such as copper and cobalt.
Original publication
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.

Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.