New synthetic technology for medicines and fine chemicals
Advertisement
A University of Tokyo research group has succeeded in synthesizing (R)- and (S)-rolipram, the active component of a medicine, in high yield with high selectivity by a catalyzed flow fine synthesis instead of the traditional batch method used in the production of 99% of medicines.
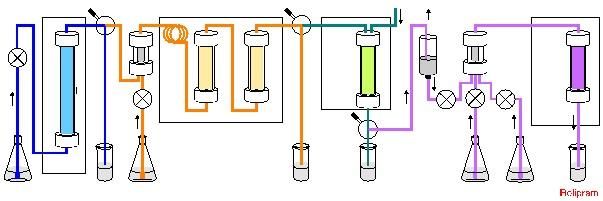
Sequential addition of starting materials enables continuous production of the final compound, rolipram.
© 2015 Shu Kobayashi.
Professor Shu Kobayashi's group at the Graduate School of Science has developed highly active immobilized catalysts (heterogeneous catalysts) and demonstrated simple and highly efficient synthesis of (R)- and (S)-rolipram by an eight-step continuous flow reaction using multiple column reactors containing the immobilized catalysts.
Currently, the active components of medicines as well as other fine chemicals are synthesized by a repeated batch reaction method, in which all starting materials are mixed in reaction vessels and the desired compounds are extracted after the all reactions have finished. In this method excess energy and operational steps are needed and a significant amount of waste is generated.
Professor Kobayashi's application of flow chemistry techniques to the production of fine chemicals using heterogeneous catalysts has resulted in simple method to synthesize (R)- and (S)-rolipram without requiring the isolation or purification of intermediates, without excess amount of energy, and without purification of products from catalysts.
Professor Kobayashi says "This new technology can be applied to not only other gamma-aminobutyric acids and medicines but also various chemicals such as flavors, agricultural chemicals, and functional materials. In the future, if this innovative catalyzed flow fine synthesis is established as an original Japanese technology, we can hope for significant development of the chemical, pharmaceutical and related industries and recovery of high skill manufacturing in Japan."





























































