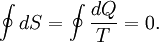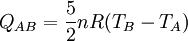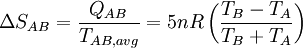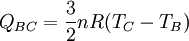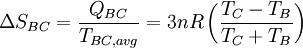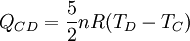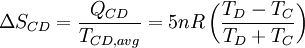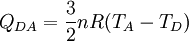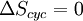To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Thermodynamic cycleA thermodynamic cycle is a series of thermodynamic processes which returns a system to its initial state. Properties depend only on the thermodynamic state and thus do not change over a cycle. Variables such as heat and work are not zero over a cycle, but rather are process dependent. The first law of thermodynamics dictates that the net heat input is equal to the net work output over any cycle. The repeating nature of the process path allows for continuous operation, making the cycle an important concept in thermodynamics. Thermodynamic cycles often use quasistatic processes to model the workings of actual devices. A thermodynamic cycle is a closed loop on a P-V diagram. A P-V diagrams X axis shows volume (V) and Y axis shows pressure (P). The area enclosed by the loop is the work (W) done by the process:
This work is equal to the balance of heat (Q) transferred into the system:
Equation (2) makes a cyclic process similar to an isothermal process: even though the internal energy changes during the course of the cyclic process, when the cyclic process finishes the system's energy is the same as the energy it had when the process began. If the cyclic process moves clockwise around the loop, then it represents a heat engine, and W will be positive. If it moves counterclockwise then it represents a heat pump, and W will be negative. Product highlight
ClassesTwo primary classes of thermodynamic cycles are power cycles and heat pump cycles. Power cycles are cycles which convert some heat input into a mechanical work output, while heat pump cycles transfer heat from low to high temperatures using mechanical work input. Cycles composed entirely of quasistatic processes can operate as power or heat pump cycles by controlling the process direction. On a pressure-volume or Temperature-entropy diagram, the clockwise and counterclockwise directions indicate power and heat pump cycles, respectively. Thermodynamic power cycles
Thermodynamic power cycles are the basis for the operation of heat engines, which supply most of the world's electric power and run almost all motor vehicles. Power cycles can be divided according to the type of heat engine they seek to model. The most common cycles that model internal combustion engines are the Otto cycle, which models gasoline engines and the Diesel cycle, which models diesel engines. Cycles that model external combustion engines include the Brayton cycle, which models gas turbines, and the Rankine cycle, which models steam turbines. For example the pressure-volume mechanical work done in the heat engine cycle, consisting of 4 thermodynamic processes, is: If no volume change happens in process 4->1 and 2->3, equation (3) simplifies to: Thermodynamic heat pump and refrigeration cycleThermodynamic heat pump and refrigeration cycles are the models for heat pumps and refrigerators. The difference between the two is that heat pumps are intended to keep a place warm and refrigerators designed to cool it. The most common refrigeration cycle is the vapor compression cycle, which models systems using refrigerants that change phase. The absorption refrigeration cycle is an alternative that absorbs the refrigerant in a liquid solution rather than evaporating it. Gas refrigeration cycles include the reversed Brayton cycle and the Linde-Hampson cycle. Regeneration in gas refrigeration allows for the liquefaction of gases. Types of thermodynamic cyclesA thermodynamic cycle can (ideally) be made out of 3 or more thermodynamic processes (typical 4). The processes can be any of these:
Some examples are as follows:
Carnot cycleThe Carnot cycle is a cycle composed of the totally reversible processes of isentropic compression and expansion and isothermal heat addition and rejection. The thermal efficiency of a Carnot cycle depends only on the temperatures in Kelvin of the two reservoirs in which heat transfer takes place, and for a power cycle is: where TL is the lowest cycle temperature and TH the highest. For Carnot refrigeration cycles the coefficient of performance for a heat pump is: and for a refrigerator the coefficient of performance is: The second law of thermodynamics limits the efficiency and COP for all cyclic devices to levels at or below the Carnot efficiency. The Stirling cycle and Ericsson cycle are two other reversible cycles that use regeneration to obtain isothermal heat transfer. Ideal cycleAn ideal cycle is constructed out of:
Otto cycleAn Otto cycle is constructed out of:
The adiabatic processes are impermeable to heat: heat flows into the loop through the left pressurizing process and some of it flows back out through the right depressurizing process, and the heat which remains does the work. Stirling cycleA Stirling cycle is like an Otto cycle, except that the adiabats are replaced by isotherms.
Heat flows into the loop through the top isotherm and the left isochore, and some of this heat flows back out through the bottom isotherm and the right isochore, but most of the heat flow is through the pair of isotherms. This makes sense since all the work done by the cycle is done by the pair of isothermal processes, which are described by Q=W. This suggests that all the net heat comes in through the top isotherm. In fact, all of the heat which comes in through the left isochore comes out through the right isochore: since the top isotherm is all at the same warmer temperature TH and the bottom isotherm is all at the same cooler temperature TC, and since change in energy for an isochore is proportional to change in temperature, then all of the heat coming in through the left isochore is cancelled out exactly by the heat going out the right isochore. State Functions and EntropyIf Z is a state function then the balance of Z remains unchanged during a cyclic process:
If entropy is defined as so that
then it can be proven that for any cyclic process, DemonstrationPart 1Draw a rectangle on a P-V diagram, such that the top and bottom are horizontal isobaric processes and the left and right are vertical isochoric processes. Such a rectangle should be made really small, so that change in temperature can be averaged out, and so that the cycle will enclose an area Δarea. Let the top left corner be labeled A, then label the rest of the corners clockwise starting from A as ABCD.
Assume that the system is a monatomic gas. Then
Process BC:
Process CD:
Process DA:
Process ABCDA (cyclic): Part 2Any loop can be broken up into a rectangular grid of differential areas. The line integral of the entire loop is equal to the sum of the line integrals of each of the constituent differential areas. Let all these integrals be done clockwise. Then any pair of adjacent differential areas will be sharing a process as a common border, but one area will add that process in one direction while the adjacent area adds that process in the reverse direction, so that process is cancelled out. Therefore all processes internal to the loop cancel each other out (see Green's theorem), and the result of the summation is equal to the line integral of the contour of the loop:
Q.E.D. ConclusionThe fact that entropy is a state function is what puts entropy on the map (the P-V diagram). References
See also
Categories: Thermodynamic cycles | Thermodynamics |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Thermodynamic_cycle". A list of authors is available in Wikipedia. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||


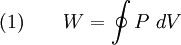 .
.
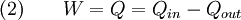 .
.



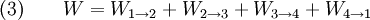
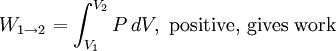
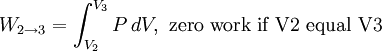
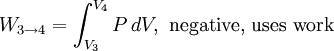
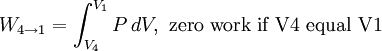
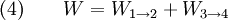
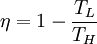
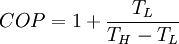
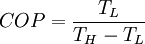
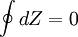 .
.
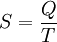
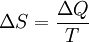 ,
,