Molecular multitools
Researchers develop method to structure surfaces using visible light
Advertisement
The functionalization of surfaces with different physical or chemical properties is a key challenge for many applications. For example, the defined structuring of a surface with hydrophobic and hydrophilic areas can be used for the separation of emulsions, like water and oil. However, the creation of user-defined surface properties is a challenge. Researches from the Max Planck Institute for Polymer Research in Mainz (MPI-P), the University of Science and Technology of China in Hefei and the University of Electronic Science and Technology in Chengdu (China) have now developed surfaces that can easily be patterned with different functionalities using visible light.
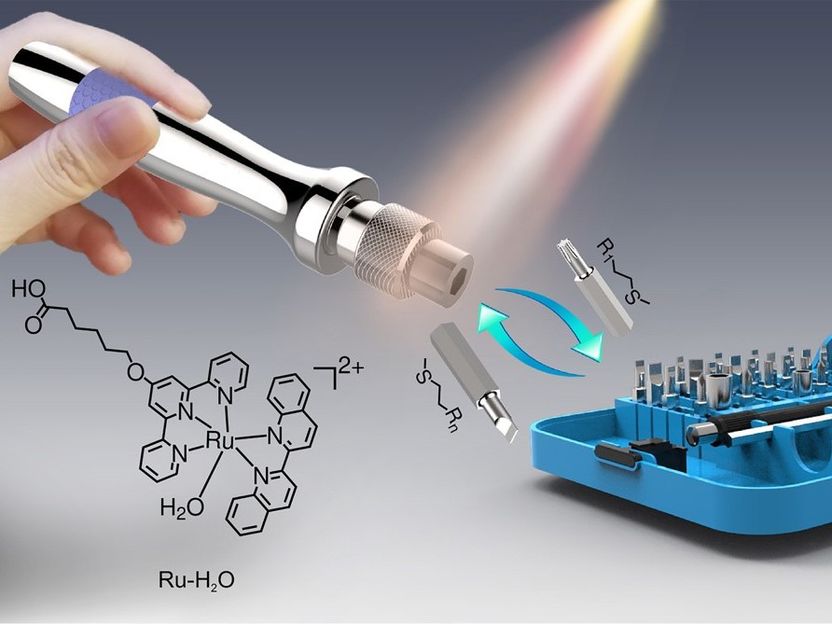
Schematic illustration of the visible-light-controlled reconfigurable surface functions. Ruthenium-complexes act as a molecular screwdriver, and thioethers with different functional groups act as molecular bits.
© MPI-P
The international team of researchers created surfaces which are coated with a molecule which has a Ruthenium-atom in its center. This molecule-complex, which is permanently attached to the surface, acts as a molecular screwdriver: “You can think of this molecule as a screwdriver, and we can attach different bits – that means molecules allowing different functionalities like wettability – to this screwdriver”, says Prof. Dr. Si Wu, group leader at the MPI-P (department of Prof. Dr. Hans-Jürgen Butt).
The attachment of such bits – here, so called thioether groups, organic molecules containing a sulfur atom – has so far been performed by chemical bonds which could not be released easily. In the past, the surface functionalities could only be removed using complicated chemical removal methods, which often destroyed not only the thioether, but also the Ruthenium complexes. In their work, the researchers showed that their molecules allow the removal of the “bits” – that means the thioether groups – by using visible light. “This is of great importance if we think of using biomolecules at the surface, which can easily be destroyed by using UV light. So in our experiment, we use visible light, which has less energy and thus doesn’t destroy biomolecules”, says Wu.
With their method, it is possible to structure surfaces in an easy way. In the dark, the whole surface area is functionalized with a desired molecule, giving for example the possibility to create hydrophobic areas. The surface is then illuminated through a shadow mask with light – this cleaves the bond between the Ruthenium complex attached to the surface and the functional thioether group. After washing the surface, the functional groups are removed at the illuminated surface areas, leaving only the non-illuminated parts.
As the Ruthenium complex is not washed away, it stays on the surface and can then – after washing – be used again to attach another bit. Thus, the surface is reconfigurable multiple times.

































































