The dynamic heart of Cu/ZnO/Al2O3 catalysts
Cu/ZnO/Al₂O₃ continuously switches between two structural states during operation — and that may be the key to its activity
Advertisement
With the aim to precisely understand its function, researchers from the Inorganic Chemistry Department and Interface Science Department of the Fritz Haber Institute, together with colleagues from the Max Planck Institute for Chemical Energy Conversion investigated the Cu/ZnO/Al2O3 catalyst system used for industrial methanol production during reaction conditions. They found that the dynamic, temperature-sensitive nature of the Cu-ZnO interaction is the key to its function – opening up new avenues for rationally improving this process.
Methanol, a chemical beacon of hope
Methanol (CH3OH) is one of the world's most important basic chemicals. It is used as a solvent, in the synthesis of plastics and paints, and as a fuel. Due to its role as a key product for the chemical industry and future climate-neutral energy, methanol is of enormous industrial and ecological importance.
Methanol production is a large-scale industrial process in which a synthesis gas consisting of carbon monoxide (CO), carbon dioxide (CO₂), and hydrogen (H₂) are converted in a reactor using catalysts. Methanol synthesis is therefore also considered a promising approach to recycling captured CO2. Since the 1960s, catalysts made of copper-zinc-aluminum oxide (Cu/ZnO/Al₂O₃) have been used for this purpose in industry. High pressure and moderate temperatures are found to favor the reaction.
However, it is still unclear how the catalysts behave in detail under different reaction conditions - which means “at work” or “operando”- and how they change and develop during the reaction. In particular, researchers are still unclear about the nature and location of the synergistic effects between Cu and ZnO, the specific nature of the active sites and their location. To put it in a nutshell, the question is: Why does the combination of Cu and ZnO exhibit such a good catalytic performance?
The current approach
Although it is known that the high activity of Cu/ZnO/Al₂O₃ catalysts comes from the synergy between Cu and ZnO, the underlying chemical processes including the interplay of catalyst components and changes of the catalyst structure under reaction conditions are not well understood.
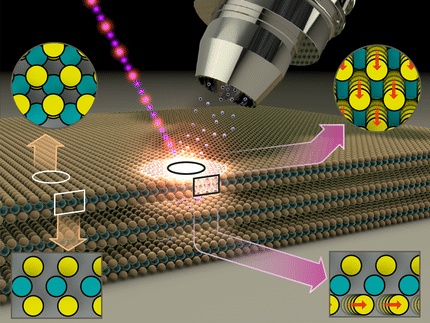
This study uses operando transmission electron microscopy (TEM) to investigate how the Cu/ZnO/Al₂O₃ catalyst develops “at work”. The researchers investigate Cu/ZnO/Al₂O₃ nanoparticles in a minute reactor, in which they can adjust the reaction conditions. In doing so, they reveal the dynamic structural changes of the Cu/ZnO/Al₂O₃ catalyst during the activation and hydrogenation of CO₂ - key processes in methanol synthesis.
The frustrated state
The experimental data show that the surface of the catalyst is highly dynamic and reversible structural transformations are driven by temperature. For instance, ZnOₓ overlayers on the catalyst surface open up at reaction temperatures above 220°C, exposing Cu surfaces for the catalytic CO₂ activation. Upon cooling, the overlayer reforms, while its thickness depends on the composition of the gas floating through the reactor.
Interestingly, the researchers hypothesized a very special state of the catalyst: a “frustrated phase transition” in which the catalyst constantly and reversibly changes its structure. Researchers call such conditions “frustrated” because they never come to rest, are virtually trapped in restlessness between the interconversion of two states, as certain physical and chemical driving forces cannot be satisfied. In the observed frustrated state, CuZn surface regions and Cu-ZnO interfacial sites on the catalyst surface continuously transform into each other while the catalyst is working. This dynamic balance is likely critical, maybe even the key parameter for the high activity and stability of the catalyst.
Conclusion
The idea behind the study was to shed light on the synergistic effects of Cu and ZnO in catalytic CO2 hydrogenation and methanol synthesis. The study answers some aspects of this question: It clearly reveals that the high performance of Cu/ZnO/Al₂O₃ catalysts is not based on a single active phase. Rather, the catalyst undergoes constant changes in its structure, whereas its activity strongly depends on the dynamic interplay between reversibly formed CuZn regions and Cu-ZnO interfaces. This “frustrated phase transition” is central to the function of the catalyst and offers new design principles for next-generation catalysts – for methanol synthesis and related processes.
Original publication
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.

Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.