Catalysts team up with textiles
Simple textiles can be used with catalysts to enable complex chemical reactions
Advertisement
In future, it will be much easier to produce some active pharmaceutical substances and chemical compounds than was the case to date. An international team working with chemists from the Max-Planck-Institut für Kohlenforschung in Mülheim an der Ruhr have immobilised various catalysts on nylon in a very simple way. Catalysts mediate between the reagents in a chemical reaction and control the process leading to the desired end product. When textile material is used as a support for the chemical auxiliaries, the reaction can proceed on a large surface thereby increasing its efficiency. One of the catalysts that the researchers used in this way plays an important role in the synthesis of a pharmaceutical agent which could only be used previously in dissolved form, making the production process very complicated and expensive. Immobilising this catalyst on fabric simplifies production considerably. This process may be expected to yield similar advantages for other chemical processes.
Functional textiles are usually understood as the textiles used to make windproof jackets, breathable footwear and particularly effective thermal underwear. However, the term could soon refer to something else – textiles which are “functionalised” with the help of organic catalysts. Working in collaboration with scientists from the Deutsches Textilforschungszentrum in Krefeld and Sungkyunkwan University in Suwon, Korea, researchers at the Max-Planck-Institut für Kohlenforschung in Mülheim an der Ruhr have developed a process for immobilising different organic catalysts on textiles with the help of ultraviolet light. The fabric thereby acts as a support for the substances on which a chemical reaction occurs.
Up to now, science has focused more on the macroscopic functionality of textiles, for example clothing, explains Ji-Woong Lee who recently completed his doctorate at the Max-Planck-Institut für Kohlenforschung under the supervision of Benjamin List, head of the Institute’s Homogenous Catalysis Group. “As opposed to this, our method can give simple textiles microscopic functionalities,” explains the Korean scientist. Together with his colleagues, Lee armed pieces of nylon with catalysts. The latter can be imagined as chemical tools which fulfil various tasks during chemical reactions.
Excellent yields, little wear and tear
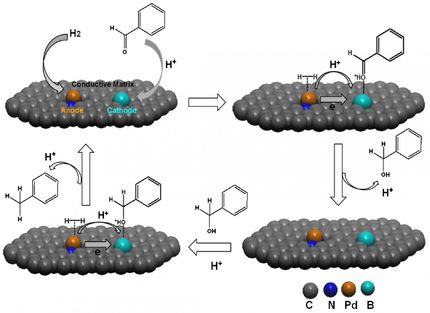
For their tests, the Mühlheim-based researchers used three organic catalysts: a base (dimethylaminopyridine, DMAP), a sulfonic acid and a catalyst which functions as both an acid and a base. The latter is used in the pharmaceuticals industry to steer a reaction to one of two products, which are chemically completely identical. The two forms have mirror-image structures, like a left and right hand, but only one variant has the desired medical effect. Up to now, the catalyst that generates this variant could only be used in dissolved form and then had to be separated again. The complicated separation process could be avoided using a catalyst immobilised on fabric.
To attach the catalysts to the nylon fibres, the chemists irradiated the textile to which a catalyst was applied with UV light for five minutes – but no longer, as this would impede the activity of the catalyst and its immobilisation on the nylon. A comparable process did not exist up to now.
The catalysts, which were practically interwoven with the fabric, displayed all of the characteristics that the chemists expect from such a system: the result of the chemical reactions which the scientists undertook with the catalyst-loaded nylon strips is impressive. All three catalysts converted around 90 percent of the source materials to the desired products. And the catalyst, which is used in the pharmaceutical industry and only generates one out of two mirror-image molecules, achieved a success rate of over 95 percent without showing any major signs of wear and tear. Ji-Woong Lee carried out several hundred test-runs and observed that the catalysts relinquished little of their functionality.
A large surface makes chemical reactions more efficient
Compared with other ways of immobilising catalysts, “organotextile catalysis” has several advantages: in particular, it provides the reagents with a larger surface than other supports, for example plastic spheres or foils – the larger the surface, the more efficiently a reaction proceeds. Moreover, nylon is flexible and very inexpensive. Dry textiles loaded with catalysts are easy to transport, which means that it is simpler to meet the requirements for some chemical processes where it is practically impossible to set up sophisticated chemical systems. For example, organotextile catalysis could help in the treatment of water in locations where people are cut off from the water supply.
“Our method enables the low-cost production of long-term functionalised textiles without causing any pollution,” says Ji-Woong Lee. He is entirely convinced that the process can be applied in several scientific areas – and industrial processes. “In addition to chemistry, these could include biology, the materials science and pharmaceutics.”
Original publication
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.

Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.