Tagging-detagging for purification of radiotracers
Advertisement
Veronique Gouverneur and colleagues from the University of Oxford and AstraZeneca, Cheshire, have developed a novel tagging-detagging strategy. This could be extremely useful for the purification of known and novel 18F-radiotracers for positron emission tomography. It is the first detagging process to rely on a C-F bond forming event and they have shown its feasibility. To do this they synthesised fluorous allylsilanes by Cross Metathesis and then fluorinated them via an Electrophilic substitution, using Selectfluor as the detagging reagent. Using this new method, both the Fluorination and the detagging occur in the last step of the sequence. The F-target can therefore be separated from the precursor using FSPE (fluorous solid phase extraction), dramatically reducing the time required to produce the pure final product.
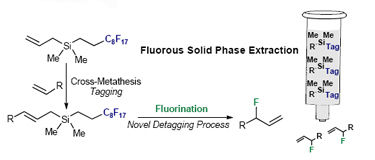
Novel Detagging process allows Fluorous Solid Phase Extraction
Their next goal will be to demonstrate that this chemistry can be done "hot", with the synthesis of known radiotracers; 'We envisage this work extending to a wider range of possible fluorodetagging strategies' Gouverneur states. 'The challenge will be to further expand the usefulness of fluorous chemistry in the context of radiochemistry.'
Original publication: Véronique Gouverneur et al., Chem. Commun., 2008.
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Extraction
Extraction is a fundamental process in the chemical laboratory that enables specific components to be isolated and concentrated from a mixture. Whether it's extracting active ingredients from natural products, removing impurities from synthesis products, or preparing analytical samples, extraction is a key step in achieving precise and efficient results in chemical research and analysis.

Topic world Extraction
Extraction is a fundamental process in the chemical laboratory that enables specific components to be isolated and concentrated from a mixture. Whether it's extracting active ingredients from natural products, removing impurities from synthesis products, or preparing analytical samples, extraction is a key step in achieving precise and efficient results in chemical research and analysis.


































































