New synthetic route to oxindoles
Advertisement
Peter Kündig and colleagues from Geneva University, Switzerland and Stephen Marsden and co-workers at the University of Leeds, in the UK, have developed an asymmetric Pd/N-heterocyclic carbene (NHC) - catalysed intramolecular -Arylation of amide enolates, containing heteroatom sustituents. This catalytic reaction produced optically active oxindole derivatives.
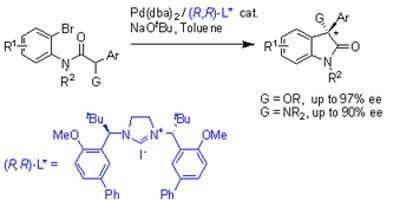
This synthetic route could be useful in the synthesis of biologically active chiral oxindoles. This work is significant as oxindoles constitute many natural products and biologically active compounds that could be developed into drugs.
Optically active oxindoles exhibit a large variety of pharmacological activity, particularly affecting neurological functions, which has led to intense activity in the asymmetric synthetic routes to prepare these compounds.
The next step is 'understanding why these particular ligands work so much better than previously tested ligands belonging to the same family (NHC's) and applying the method to more complex bioactive molecules,' said Kündig.
Original publication: E. Peter Kündig et al., Chem. Commun., 2008.
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.

Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.