To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Free entropyA thermodynamic free entropy is an entropic thermodynamic potential analogous to the free energy. Also know as a Massieu, Planck, or Massieu-Planck potentials (or functions), or (rarely) free information. In statistical mechanics, free entropies frequently appear as the logarithm of a partition function. In mathematics, free entropy is the generalization of entropy defined in free probability. A free entropy is generated by a Legendre transform of the entropy. The different potentials correspond to different constraints to which the system may be subjected. The most common examples are:
Note that the use of the terms "Massieu" and "Planck" for explicit Massieu-Planck potentials are somewhat obscure and ambiguous. In particular "Planck potential" has alternative meanings. The most standard notation for an entropic potential is ψ, used by both Planck and Schrodinger. (Note that Gibbs used ψ to denote the free energy.) Free entropies where invented by Massieu in 1869, and actually predate Gibb's free energy (1875). Product highlight
Dependence of the potentials on the natural variablesEntropy
By the definition of a total differential,
From the equations of state,
The differentials in the above equation are all of extensive variables, so they may be integrated to yield
Massieu potential \ Helmholtz free entropyStarting over at the definition of Φ and taking the total differential, we have via a Legendre transform (and the chain rule)
The above differentials are not all of extensive variables, so the equation may not be directly integrated. From dΦ we see that
If reciprocal variables are not desired,[3]
Planck potential \ Gibbs free entropyStarting over at the definition of Ξ and taking the total differential, we have via a Legendre transform (and the chain rule)
The above differentials are not all of extensive variables, so the equation may not be directly integrated. From dΞ we see that
If reciprocal variables are not desired,[3]
References
Categories: Thermodynamics | Thermodynamic entropy |
||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Free_entropy". A list of authors is available in Wikipedia. |


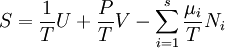
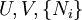
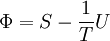
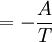
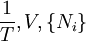
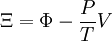
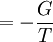
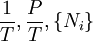



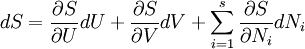 .
.
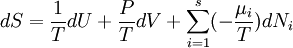 .
.
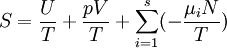 .
.
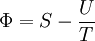
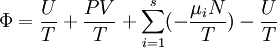
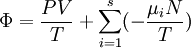
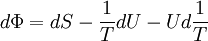 ,
,
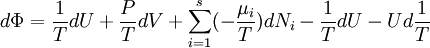 ,
,
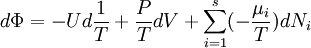 .
.
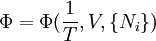 .
.
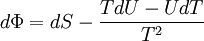 ,
,
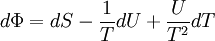 ,
,
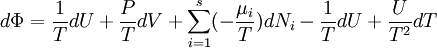 ,
,
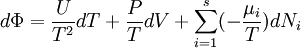 ,
,
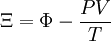
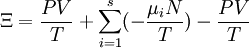
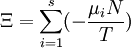
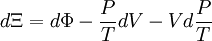
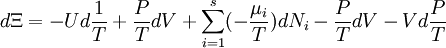
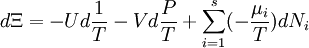 .
.
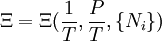 .
.
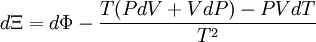 ,
,
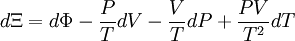 ,
,
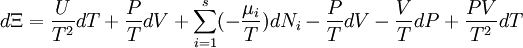 ,
,
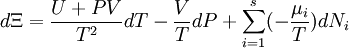 ,
,


