The AETX files: Work to unravel widespread bald eagle fatalities gathers pace
Scientists succeed in totally synthesizing eagle killer venom
Advertisement
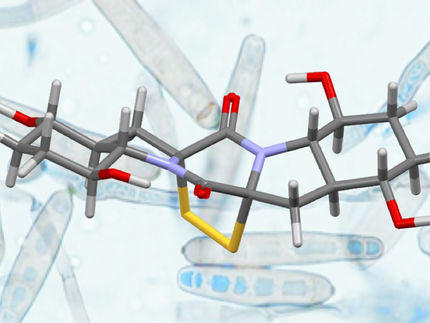
This March, the discovery of the toxic natural substance aetocthonotoxin (AETX) attracted huge media attention. In a study published in Science, pharmacists from Halle University proved the role of this substance in triggering mass species extinctions among American bald eagles and other birds of prey. Now, in another first, natural product chemists at the Leibniz Institute of Plant Biochemistry (IPB) have broken new ground in completely synthesizing aetocthonotoxin. This will pave the way to produce the novel toxin in sufficient quantities to study both its biological activity and its impact on human health in more detail.
The American heraldic animal is under massive threat from the nerve toxin AETX.
pixabay.com
Aetocthonotoxin (AETX) is an alkaloid that includes several structural peculiarities of stand-out rarity, even among what are sometimes very complex natural products. The substance comprises several ring structures, to which five bromine atoms are coupled. "Creating compounds as complex as this in a test tube is a major challenge for synthetic chemists," commented the study director, Professor Bernhard Westermann. All such hurdles, however, were successfully overcome for AETX, despite the fact that only five reaction steps are involved in synthesizing the toxin. Testament to the design expertise of participating scientists from IPB and MLU.
Alkaloids form a group of plant secondary metabolites featuring great structural diversity and often comprising several carbon rings that contain at least one nitrogen atom. Most are poisonous and very bitter-tasting. Even so, many alkaloid-bearing plants such as belladonna, the opium poppy, arnica and camomile have often been harnessed for medicinal purposes over thousands of years. Incidentally, the patron saint of alkaloids comes from Halle: Namely, the owner of the Löwenapotheke, Carl Friedrich Wilhelm Meißner, who coined the term ‘alkaloid’ in 1819.
Ever since coming into existence in 1958, the IPB has intensively researched the synthesis and biosynthesis of key alkaloid substances such as morphine, cocaine, THC and ergot alkaloids. Founding director and Leopoldina president Kurt Mothes invited scientists from all over the world to Halle to provide an optimal forum for discussing this interesting substance group. It was on his initiative that International Conferences on the Biochemistry and Physiology of Alkaloids were held in the city in 1960, 1964 and 1969. With AETX synthesis, IPB natural product chemists cement the historic paths of alkaloid research at the institute. Scope to produce the active ingredient synthetically now opens up new potential applications. Because: What separates poison and medicine is often close to imperceptible. Thanks to chemical modifications, AETX compounds with desirable pharmacological properties can often be produced.
Background: AETX is produced from the Aekthonotoxin hydrillicola cyanobacterium, which is found on the leaves of the freshwater plant growing invasively in North America Hydrilla verticillata . The toxin travels through the food chain, starting with herbivores, such as snails and waterfowl, and ending with birds of prey, such as eagles and kites. In the bald eagle, the substance triggers a previously unknown neurodegenerative and fatal disease: vacuolar myelinopathy. The tell-tale sign of the disease is a breakdown of the insulating myelin layers around the nerve fibers of the brain and spinal cord. Whether the toxin is equally harmful to mammals remains unclear.
Original publication
Other news from the department science
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.

Topic world Synthesis
Chemical synthesis is at the heart of modern chemistry and enables the targeted production of molecules with specific properties. By combining starting materials in defined reaction conditions, chemists can create a wide range of compounds, from simple molecules to complex active ingredients.