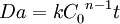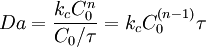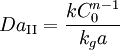To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Damköhler numbersThe Damköhler numbers (Da) are dimensionless numbers used in chemical engineering to relate chemical reaction timescale to other phenomena occurring in a system. It is named after German chemist Gerhard Damköhler, 1908-1944. Product highlightThere are several Damköhler numbers, and their definition varies according to the system under consideration. For a general chemical reaction A → B of n-th order, the Damköhler number is defined as where:
and it represents a dimensionless reaction time. It provides a quick estimate of the degree of conversion (X) that can be achieved in continous flow reactors. Generally, if Da<0.1 then X<0.1 similarly if Da>10 then X>0.9 In continuous or semibatch chemical processes, the general definition of the Damköhler number is defined as:
For example, in a continuous reactor, the Damköhler number is: where τ is the mean residence time or space time. In reacting systems that include also interphase mass transport, the second Damköhler number (DaII) is defined as the ratio of the chemical reaction rate to the mass transfer rate where
Categories: Chemical engineering | Chemical kinetics |
||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Damköhler_numbers". A list of authors is available in Wikipedia. |